
In 1953 Stanley Miller, a graduate student at the University of Chicago, took two flasks—one containing a little water to represent a primeval ocean, the other holding a mixture of methane, ammonia and hydrogen sulphide gases to represent the Earth’s early atmosphere—connected them with rubber tubes and introduced some electrical sparks as a stand-in for lightning. After a few days, the water in the flasks had turned green and yellow in a hearty broth of amino acids, fatty acids, sugars and other organic compounds. “If God didn’t do it this way,” observed Miller’s delighted supervisor, the Nobel laureate Harold Urey, “He missed a good bet.”
Press reports of the time made it sound as if about all that was needed now was for somebody to give the flasks a good shake and life would crawl out. As time has shown, it wasn’t nearly so simple. Despite half a century of further study, we are no nearer to synthesizing life today than we were in 1953—and much further away from thinking we can. Scientists are now pretty certain that the early atmosphere was nothing like as primed for development as Miller and Urey’s gaseous stew, but rather was a much less reactive blend of nitrogen and carbon dioxide. Repeating Miller’s experiments with these more challenging inputs has so far produced only one fairly primitive amino acid. At all events, creating amino acids is not really the problem. The problem is proteins.
Proteins are what you get when you string amino acids together, and we need a lot of them. No-one really knows, but there may be as many as a million types of protein in the human body, and each one is a little miracle. By all the laws of probability proteins shouldn’t exist. To make a protein you need to assemble amino acids (which I am obliged by long tradition to refer to here as “the building blocks of life”) in a particular order, in much the same way that you assemble letters in a particular order to spell a word. The problem is that words in the amino-acid alphabet are often exceedingly long. To spell “collagen,” the name of a common type of protein, you need to arrange eight letters in the right order. To make collagen, you need to arrange 1,055 amino acids in precisely the right sequence. But—and here’s an obvious but crucial point—you don’t make it. It makes itself, spontaneously, without direction, and this is where the unlikelihoods come in.
The chances of a 1,055-sequence molecule like collagen spontaneously self-assembling are, frankly, nil. It just isn’t going to happen. To grasp what a long shot its existence is, visualize a standard Las Vegas slot machine but broadened greatly—to about 27 metres, to be precise—to accommodate 1,055 spinning wheels instead of the usual three or four, and with twenty symbols on each wheel (one for each common amino acid)1 How long would you have to pull the handle before all 1,055 symbols came up in the right order? Effectively, for ever. Even if you reduced the number of spinning wheels to 200, which is actually a more typical number of amino acids for a protein, the odds against all 200 coming up in a prescribed sequence are 1 in 10260 (that is a 1 followed by 260 zeros). That in itself is a larger number than all the atoms in the universe.
Proteins, in short, are complex entities. Haemoglobin is only 146 amino acids long, a runt by protein standards, yet even it offers 10190 possible amino-acid combinations, which is why it took the Cambridge University chemist Max Perutz twenty-three years—a career, more or less—to unravel it. For random events to produce even a single protein would seem a stunning improbability—like a whirlwind spinning through a junkyard and leaving behind a fully assembled jumbo jet, in the colourful simile of the astronomer Fred Hoyle.

Stanley Miller of the University of Chicago in 1953, soon after announcing an experiment that seemed to offer the possibility of life in a test tube. The hope proved decidedly premature. (credit 19.2)
Yet we are talking about several hundred thousand types of protein, perhaps a million, each unique and each, as far as we know, vital to the maintenance of a sound and happy you. And it goes on from there. To be of use, a protein must not only assemble amino acids in the right sequence, it must then engage in a kind of chemical origami and fold itself into a very specific shape. Even having achieved this structural complexity, a protein is no good to you if it can’t reproduce itself, and proteins can’t. For this you need DNA. DNA is a whiz at replicating—it can make a copy of itself in seconds—but can do virtually nothing else. So we have a paradoxical situation. Proteins can’t exist without DNA and DNA has no purpose without proteins. Are we to assume, then, that they arose simultaneously with the purpose of supporting each other? If so: wow.
And there is more still. DNA, proteins and the other components of life couldn’t prosper without some sort of membrane to contain them. No atom or molecule has ever achieved life independently. Pluck any atom from your body and it is no more alive than is a grain of sand. It is only when they come together within the nurturing refuge of a cell that these diverse materials can take part in the amazing dance that we call life. Without the cell, they are nothing more than interesting chemicals. But without the chemicals, the cell has no purpose. As Davies puts it, “If everything needs everything else, how did the community of molecules ever arise in the first place?” It is rather as if all the ingredients in your kitchen somehow got together and baked themselves into a cake—but a cake that could moreover divide when necessary to produce more cakes. It is little wonder that we call it the miracle of life. It is also little wonder that we have barely begun to understand it.
So what accounts for all this wondrous complexity? Well, one possibility is that perhaps it isn’t quite—not quite—so wondrous as at first it seems. Take those amazingly improbable proteins. The wonder we see in their assembly comes in assuming that they arrived on the scene fully formed. But what if the protein chains didn’t assemble all at once? What if, in the great slot machine of creation, some of the wheels could be held, as a gambler might hold a number of promising cherries? What if, in other words, proteins didn’t suddenly burst into being, but evolved?
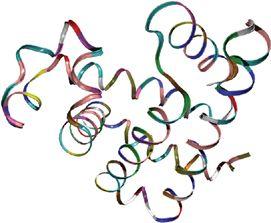
Computer model showing the tortuous—but crucially specific—ribbons and curls that make up a protein’s shape. To perform its function, a protein (this one is myoglobin, which is found in muscle tissue) must have exactly the right components correctly folded into exactly the right shape. (credit 19.3)
Imagine if you took all the components that make up a human being—carbon, hydrogen, oxygen and so on—and put them in a container with some water, gave it a vigorous stir and out stepped a completed person. That would be amazing. Well, that’s essentially what Hoyle and others (including many ardent creationists) argue when they suggest that proteins spontaneously formed all at once. They didn’t—they can’t have. As Richard Dawkins argues in The Blind Watchmaker, there must have been some kind of cumulative selection process that allowed amino acids to assemble in chunks. Perhaps two or three amino acids linked up for some simple purpose and then after a time bumped into some other similar small cluster and in so doing “discovered” some additional improvement.
Chemical reactions of the sort associated with life are actually something of a commonplace. It may be beyond us to cook them up in a lab, à la Stanley Miller and Harold Urey, but the universe does it readily enough. Lots of molecules in nature get together to form long chains calledpolymers. Sugars constantly assemble to form starches. Crystals can do a number of lifelike things—replicate, respond to environmental stimuli, take on a patterned complexity. They’ve never achieved life itself, of course, but they demonstrate repeatedly that complexity is a natural, spontaneous, entirely reliable event. There may or may not be a great deal of life in the universe at large, but there is no shortage of ordered self-assembly, in everything from the transfixing symmetry of snowflakes to the comely rings of Saturn.

Snowflakes, like all crystals, are marvels of spontaneous design and a reminder that ordered complexity is commonplace in the universe. (credit 19.4)
So powerful is this natural impulse to assemble that many scientists now believe that life may be more inevitable than we think—that it is, in the words of the Belgian biochemist and Nobel laureate Christian de Duve, “an obligatory manifestation of matter, bound to arise wherever conditions are appropriate.” De Duve thought it likely that such conditions would be encountered perhaps a million times in every galaxy.
Certainly there is nothing terribly exotic in the chemicals that animate us. If you wished to create another living object, whether a goldfish or a head of lettuce or a human being, you would need really only four principal elements, carbon, hydrogen, oxygen and nitrogen, plus small amounts of a few others, principally sulphur, phosphorus, calcium and iron. Put these together in three dozen or so combinations to form some sugars, acids and other basic compounds and you can build anything that lives. As Dawkins notes: “There is nothing special about the substances from which living things are made. Living things are collections of molecules, like everything else.”
The bottom line is that life is amazing and gratifying, perhaps even miraculous, but hardly impossible—as we repeatedly attest with our own modest existences. To be sure, many of the fine details of life’s beginnings remain pretty imponderable. Every scenario you have ever read concerning the conditions necessary for life involves water—from the “warm little pond” where Darwin supposed life began to the bubbling sea vents that are now the most popular candidates for life’s beginnings—but all this overlooks the fact that to turn monomers into polymers (which is to say, to begin to create proteins) involves a type of reaction known to biology as “dehydration linkages.” As one leading biology text puts it, with perhaps just a tiny hint of discomfort, “Researchers agree that such reactions would not have been energetically favorable in the primitive sea, or indeed in any aqueous medium, because of the mass action law.” It is a little like putting sugar in a glass of water and having it become a cube. It shouldn’t happen, but somehow in nature it does. The actual chemistry of all this is a little arcane for our purposes here, but it is enough to know that if you make monomers wet they don’t turn into polymers—except when creating life on the Earth. How and why it happens then and not otherwise is one of biology’s great unanswered questions.

One of the biggest surprises in the earth sciences in recent decades was discovering just how early in Earth’s history life arose. Well into the 1950s, it was thought that life was less than six hundred million years old. By the 1970s, a few adventurous souls felt that maybe it went back 2.5 billion years. But the present date of 3.85 billion years is stunningly early. The Earth’s surface didn’t become solid until about 3.9 billion years ago.

A meteor streaks across the sky of Wales in 2003. Most meteors burn up harmlessly in the atmosphere. Those that strike the ground become known as meteorites. (credit 19.5)
“We can only infer from this rapidity that it is not ‘difficult’ for life of bacterial grade to evolve on planets with appropriate conditions,” Stephen Jay Gould observed in the New York Times in 1996. Or as he put it elsewhere, it is hard to avoid the conclusion that “life, arising as soon as it could, was chemically destined to be.”
Life emerged so swiftly, in fact, that some authorities think it must have had help—perhaps a good deal of help. The idea that earthly life might have arrived from space has a surprisingly long and even occasionally distinguished history. The great Lord Kelvin himself raised the possibility as long ago as 1871 at a meeting of the British Association for the Advancement of Science, when he suggested that “the germs of life might have been brought to the earth by some meteorite.” But it remained little more than a fringe notion until one Sunday in September 1969 when tens of thousands of Australians were startled by a series of sonic booms and the sight of a fireball streaking from east to west across the sky. The fireball made a strange crackling sound as it passed and left behind a smell that some likened to methylated spirits and others described as just awful.

A prized chunk from the celebrated fireball that exploded over Murchison, Australia, in 1969. (credit 19.6)
The fireball exploded above Murchison, a town of six hundred people in the Goulburn Valley north of Melbourne, and came raining down in chunks, some weighing over 5 kilograms. Fortunately, no-one was hurt. The meteorite was of a rare type known as a carbonaceous chondrite, and the townspeople helpfully collected and brought in some 90 kilograms of it. The timing could hardly have been better. Less than two months earlier, the Apollo 11 astronauts had returned to Earth with a bag full of lunar rocks, so labs throughout the world were geared up—indeed, clamouring—for rocks of extraterrestrial origin.
The Murchison meteorite was found to be 4.5 billion years old, and it was studded with amino acids—seventy-four types in all, eight of which are involved in the formation of earthly proteins. In late 2001, more than thirty years after it crashed, a team at the Ames Research Center in California announced that the Murchison rock also contained complex strings of sugars called polyols, which had not been found off the Earth before.
A few other carbonaceous chondrites have strayed into the Earth’s path since 1969—one that landed near Tagish Lake in Canada’s Yukon in January 2000 was seen over large parts of North America—and they have likewise confirmed that the universe is actually rich in organic compounds. Halley’s comet, it is now thought, is about 25 per cent organic molecules. Get enough of those crashing into a suitable place—Earth, for instance—and you have the basic elements you need for life.
There are two problems with notions of panspermia, as extraterrestrial theories are known. The first is that it doesn’t answer any questions about how life arose, but merely moves responsibility for it elsewhere. The other is that panspermia tends sometimes to excite even the most respectable adherents to levels of speculation that can be safely called imprudent. Francis Crick, co-discoverer of the structure of DNA, and his colleague Leslie Orgel have suggested that Earth was “deliberately seeded with life by intelligent aliens,” an idea that Gribbin calls “at the very fringe of scientific respectability”—or, put another way, a notion that would be considered wildly lunatic were it voiced by anyone other than a Nobel laureate. Fred Hoyle and his colleague Chandra Wickramasinghe further eroded enthusiasm for panspermia by suggesting, as noted inChapter 3, that outer space brought us not only life but also many diseases such as flu and bubonic plague, ideas that were easily disproved by biochemists.

The British astrophysicist Sir Fred Hoyle, immortalized (a touch unexpectedly) in a mosaic created for Britain’s National Gallery in 1952. Hoyle thought that many earthly diseases came from outer space. (credit 19.7)
Whatever prompted life to begin, it happened just once. That is the most extraordinary fact in biology, perhaps the most extraordinary fact we know. Everything that has ever lived, plant or animal, dates its beginnings from the same primordial twitch. At some point in an unimaginably distant past some little bag of chemicals fidgeted to life. It absorbed some nutrients, gently pulsed, had a brief existence. This much may have happened before, perhaps many times. But this ancestral packet did something additional and extraordinary: it cleaved itself and produced an heir. A tiny bundle of genetic material passed from one living entity to another, and has never stopped moving since. It was the moment of creation for us all. Biologists sometimes call it the Big Birth.
“Wherever you go in the world, whatever animal, plant, bug or blob you look at, if it is alive, it will use the same dictionary and know the same code. All life is one,” says Matt Ridley. We are all the result of a single genetic trick handed down from generation to generation over nearly four billion years, to such an extent that you can take a fragment of human genetic instruction and patch it into a faulty yeast cell and the yeast cell will put it to work as if it were its own. In a very real sense, it is its own.
The dawn of life—or something very like it—sits on a shelf in the office of a friendly isotope geochemist named Victoria Bennett in the Earth Sciences building of the Australian National University in Canberra. An American, Ms. Bennett came to the ANU from California on a two-year contract in 1989 and has been there ever since. When I visited her, in late 2001, she handed me a modestly hefty hunk of rock composed of thin alternating stripes of white quartz and a grey-green material called clinopyroxene. The rock came from Akilia Island in Greenland, where unusually ancient rocks were found in 1997. The rocks are 3.85 billion years old and represent the oldest marine sediments ever found.
“We can’t be certain that what you are holding once contained living organisms because you’d have to pulverize it to find out,” Bennett told me. “But it comes from the same deposit where the oldest life was excavated, so it probably had life in it.” Nor would you find actual fossilized microbes, however carefully you searched. Any simple organisms, alas, would have been baked away by the processes that turned ocean mud to stone. Instead, what we would see if we crunched up the rock and examined it microscopically would be the chemical residues that the organisms left behind—carbon isotopes and a type of phosphate called apatite, which together provide strong evidence that the rock once contained colonies of living things. “We can only guess what the organism might have looked like,” Bennett said. “It was probably about as basic as life can get—but it was life nonetheless. It lived. It propagated.”
And eventually it led to us.
If you are into very old rocks, and Ms. Bennett indubitably is, the ANU has long been a prime place to be. This is largely thanks to the ingenuity of a man named Bill Compston, who is now retired but in the 1970s built the world’s first Sensitive High Resolution Ion Micro Probe—or SHRIMP, as it is more affectionately known from its initial letters. This is a machine that measures the decay rate of uranium in tiny minerals called zircons. Zircons appear in most rocks apart from basalts and are extremely durable, surviving every natural process but subduction. Most of the Earth’s crust has been slipped back into the interior at some point, but just occasionally—in Western Australia and Greenland, for example—geologists have found outcrops of rocks that have remained always at the surface. Compston’s machine allowed such rocks to be dated with unparalleled precision. The prototype SHRIMP was built and machined in the Earth Sciences Department’s own workshops, and looked like something that had been built from spare parts on a budget, but it worked great. On its first formal test, in 1982, it dated the oldest thing ever found—a 4.3-billion-year-old rock from Western Australia.
“It caused quite a stir at the time,” Bennett told me, “to find something so important so quickly with brand-new technology.”
She took me down the hall to see the current model, SHRIMP II. It was a big, heavy piece of stainless-steel apparatus, perhaps 3.5 metres long and 1.5 metres high, and as solidly built as a deep-sea probe. At a console in front of it, keeping an eye on ever-changing strings of figures on a screen, was a man named Bob from Canterbury University in New Zealand. He had been there since 4 a.m., he told me. It was just after 9 a.m. and Bob had the machine until noon. SHRIMP II runs twenty-four hours a day; there are that many rocks to date. Ask a pair of geochemists how something like this works, and they will start talking about isotopic abundances and ionization levels with an enthusiasm that is more endearing than fathomable. The upshot of it, however, was that the machine, by bombarding a sample of rock with streams of charged atoms, is able to detect subtle differences in the amounts of lead and uranium in the zircon samples, by which means the age of rocks can be accurately adduced. Bob told me that it takes about seventeen minutes to read one zircon and it is necessary to read dozens from each rock to make the data reliable. In practice, the process seemed to involve about the same level of scattered activity, and about as much stimulation, as a trip to a launderette. Bob seemed very happy, however; but then, people from New Zealand very generally do.
The Earth Sciences compound was an odd combination of things—part office, part lab, part machine shed. “We used to build everything here,” she said. “We even had our own glassblower, but he’s retired. But we still have two full-time rock crushers.” She caught my look of mild surprise. “We get through a lot of rocks. And they have to be very carefully prepared. You have to make sure there is no contamination from previous samples—no dust or anything. It’s quite a meticulous process.” She showed me the rock-crushing machines, which were indeed pristine, though the rock crushers had apparently gone for coffee. Beside the machines were large boxes containing rocks of all shapes and sizes. They do indeed get through a lot of rocks at the ANU.
Back in Bennett’s office after our tour, I noticed hanging on her wall a poster giving an artist’s colourfully imaginative interpretation of the Earth as it might have looked 3.5 billion years ago, just when life was getting going, in the ancient period known to earth science as the Archaean. The poster showed an alien landscape of huge, very active volcanoes, and a steamy, copper-coloured sea beneath a harsh red sky. Stromatolites, a kind of bacterial rock, filled the shallows in the foreground. It didn’t look like a very promising place to create and nurture life. I asked her if the painting was accurate.
“Well, one school of thought says it was actually cool then because the sun was much weaker.” (I later learned that biologists, when they are feeling jocose, refer to this as “the Chinese restaurant problem”—because we had a dim sun.) “Without an atmosphere ultraviolet rays from the sun, even from a weak sun, would have tended to break apart any incipient bonds made by molecules. And yet right there“—she tapped the stromatolites—”you have organisms almost at the surface. It’s a puzzle.”

Earth as it might have appeared in its Archaean infancy 3.5 billion years ago, when the Moon was much closer, volcanic eruptions commonplace (because of the thinness of the crust), meteor impacts routine and the air thick with acidic vapours. Remarkably, it was in such an unpromising environment that life first got going. (credit 19.8)
“So we don’t know what the world was like back then?”
“Mmmm,” she agreed thoughtfully.
“Either way it doesn’t seem very conducive to life.”
She nodded amiably. “But there must have been something that suited life. Otherwise we wouldn’t be here.”
It certainly wouldn’t have suited us. If you were to step from a time machine into that ancient Archaean world, you would very swiftly scamper back inside, for there was no more oxygen to breathe on the Earth back then than there is on Mars today. It was also full of noxious vapours from hydrochloric and sulphuric acids powerful enough to eat through clothing and blister skin. Nor would it have provided the clean and glowing vistas depicted in the poster in Victoria Bennett’s office. The chemical stew that was the atmosphere then would have allowed little sunlight to reach the Earth’s surface. What little you could see would be illumined only briefly by bright and frequent lightning flashes. In short, it was the Earth, but an Earth we wouldn’t recognize as our own.
Anniversaries were few and far between in the Archaean world. For two billion years bacterial organisms were the only forms of life. They lived, they reproduced, they swarmed, but they didn’t show any particular inclination to move on to another, more challenging level of existence. At some point in the first billion years of life, cyano-bacteria, or blue-green algae, learned to tap into a freely available resource—the hydrogen that exists in spectacular abundance in water. They absorbed water molecules, supped on the hydrogen and released the oxygen as waste, and in so doing invented photosynthesis. As Margulis and Sagan note, photosynthesis is “undoubtedly the most important single metabolic innovation in the history of life on the planet”—and it was invented not by plants but by bacteria.
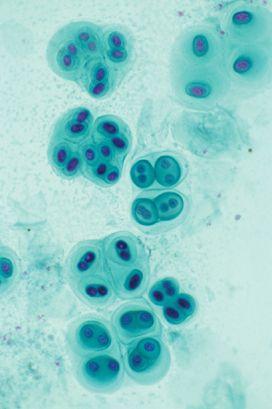
A light micrograph showing a living colony of cyanobacteria, a type of blue-green algae whose small-scale persistence transformed the planet. (credit 19.9)
As cyanobacteria proliferated the world began to fill with O2, to the consternation of those organisms that found it poisonous—which in those days was all of them. In an anaerobic (or non-oxygen-using) world, oxygen is extremely poisonous. Our white blood cells actually use oxygen to kill invading bacteria. That oxygen is fundamentally toxic often comes as a surprise to those of us who find it so convivial to our well-being, but that is only because we have evolved to exploit it. To other things it is a terror. It is what turns butter rancid and makes iron rust. Even we can tolerate it only up to a point. The oxygen level in our cells is only about a tenth the level found in the atmosphere.
The new oxygen-using organisms had two advantages. Oxygen was a more efficient way to produce energy, and it vanquished competitor organisms. Some retreated into the oozy, anaerobic world of bogs and lake bottoms. Others did likewise but then later (much later) migrated to the digestive tracts of beings like you and me. Quite a number of these primeval entities are alive inside your body right now, helping to digest your food, but abhorring even the tiniest hint of O2. Untold number of others failed to adapt and died.
The cyanobacteria were a runaway success. At first, the extra oxygen they produced didn’t accumulate in the atmosphere, but combined with iron to form ferric oxides, which sank to the bottom of primitive seas. For millions of years, the world literally rusted—a phenomenon vividly recorded in the banded iron deposits that provide so much of the world’s iron ore today. For many tens of millions of years not a great deal more than this happened. If you went back to that early Proterozoic world you wouldn’t find many signs of promise for the Earth’s future life. Perhaps here and there in sheltered pools you’d encounter a film of living scum or a coating of glossy greens and browns on shoreline rocks, but otherwise life remained invisible.

Spirulina, another of the many types of modern cyanobacteria, consists of individual algal cells strung together to form curling strands. (credit 19.10)
But about 3.5 billion years ago something more emphatic became apparent. Wherever the seas were shallow, visible structures began to appear. As they went through their chemical routines, the cyanobacteria became very slightly tacky, and that tackiness trapped micro-particles of dust and sand, which became bound together to form slightly weird but solid structures—the stromatolites that featured in the shallows of the poster on Victoria Bennett’s office wall. Stromatolites came in various shapes and sizes. Sometimes they looked like enormous cauliflowers, sometimes like fluffy mattresses (stromatolite comes from the Greek for mattress); sometimes they came in the form of columns, rising tens of metres above the surface of the water—on occasion as high as 100 metres. In all their manifestations, they were a kind of living rock, and they represented the world’s first co-operative venture, with some varieties of primitive organism living just at the surface and others living just underneath, each taking advantage of conditions created by the other. The world had its first ecosystem.
For many years, scientists knew about stromatolites from fossil formations, but in 1961 they got a real surprise with the discovery of a community of living stromatolites at Shark Bay on the remote northwest coast of Australia. This was most unexpected—so unexpected, in fact, that it was some years before scientists realized quite what they had found. Today, however, Shark Bay is a tourist attraction—or at least as much of a tourist attraction as a place hundreds of miles from anywhere much and dozens of miles from anywhere at all can ever be. Boardwalks have been built out into the bay so that visitors can stroll over the water to get a good look at the stromatolites, quietly respiring just beneath the surface. They are lustreless and grey and look, as I recorded in an earlier book, like very large cow-pats. But it is a curiously giddying moment to find yourself staring at living remnants of the Earth as it was 3.5 billion years ago. As Richard Fortey has put it: “This is truly time travelling, and if the world were attuned to its real wonders this sight would be as well-known as the pyramids of Giza.” Although you’d never guess it, these dull rocks swarm with life, with an estimated (well, obviously estimated) three billion individual organisms on every square yard of rock. Sometimes when you look carefully you can see tiny strings of bubbles rising to the surface as they give up their oxygen. In two billion years such tiny exertions raised the level of oxygen in the Earth’s atmosphere to 20 per cent, preparing the way for the next, more complex chapter in life’s history.
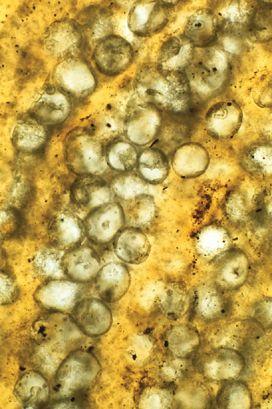
Fossilized cyanobacteria of a type that vanished 38 million years ago. For half of Earth’s long history, such simple organisms were the planet’s supreme biological achievement. (credit 19.11)
It has been suggested that the cyanobacteria at Shark Bay are perhaps the most slowly evolving organisms on Earth, and certainly now they are among the rarest. Having prepared the way for more complex life forms, they were then grazed out of existence nearly everywhere by the very organisms whose existence they had made possible. (They exist at Shark Bay because the waters are too saline for the creatures that would normally feast on them.)
One reason life took so long to grow complex was that the world had to wait until the simpler organisms had oxygenated the atmosphere sufficiently. “Animals could not summon up the energy to work,” as Fortey has put it. It took about two billion years, roughly 40 per cent of Earth’s history, for oxygen levels to reach more or less modern levels of concentration in the atmosphere. But once the stage was set, and apparently quite suddenly, an entirely new type of cell arose—one containing a nucleus and other little bodies collectively called organelles (from a Greek word meaning “little tools”). The process is thought to have started when some blundering or adventuresome bacterium either invaded or was captured by some other bacterium and it turned out that this suited them both. The captive bacterium became, it is thought, a mitochondrion. Thismitochondrial invasion (or endosymbiotic event, as biologists like to term it) made complex life possible. (In plants a similar invasion produced chloroplasts, which enable plants to photosynthesize.)
Mitochondria manipulate oxygen in a way that liberates energy from foodstuffs. Without this niftily facilitating trick, life on Earth today would be nothing more than a sludge of simple microbes. Mitochondria are very tiny—you could pack a billion into the space occupied by a grain of sand—but also very hungry. Almost every nutriment you absorb goes to feeding them.
We couldn’t live for two minutes without them, yet even after a billion years mitochondria behave as if they think things might not work out between us. They maintain their own DNA, RNA and ribosomes. They reproduce at a different time from their host cells. They look like bacteria, divide like bacteria and sometimes respond to antibiotics in the way bacteria do. They don’t even speak the same genetic language as the cell in which they live. In short, they keep their bags packed. It is like having a stranger in your house, but one who has been there for a billion years.
The new type of cells are known as eukaryotes (meaning “truly nucleated”), as contrasted with the old type, which are known as prokaryotes (“pre-nucleated”), and they seem to have arrived suddenly in the fossil record. The oldest eukaryotes yet known, called Grypania, were discovered in iron sediments in Michigan in 1992. Such fossils have been found just once and then no more are known for 500 million years.
Earth had taken its first step towards becoming a truly interesting planet. Compared with the new eukaryotes the old prokaryotes were little more than “bags of chemicals,” to borrow from the British geologist Stephen Drury. Eukaryotes were bigger—eventually as much as ten thousand times bigger—than their simpler cousins, and could carry as much as a thousand times more DNA. Gradually, thanks to these breakthroughs, life became complex and created two types of organism—those that expel oxygen (like plants) and those that take it in (like you and me).
Single-celled eukaryotes were once called protozoa (“pre-animals”), but that term is increasingly disdained. Today the common term for them is protists. Compared with the bacteria that had gone before, these new protists were wonders of design and sophistication. The simple amoeba, just one cell big and without any ambitions but to exist, contains 400 million bits of genetic information in its DNA—enough, as Carl Sagan noted, to fill eighty books of 500 pages.
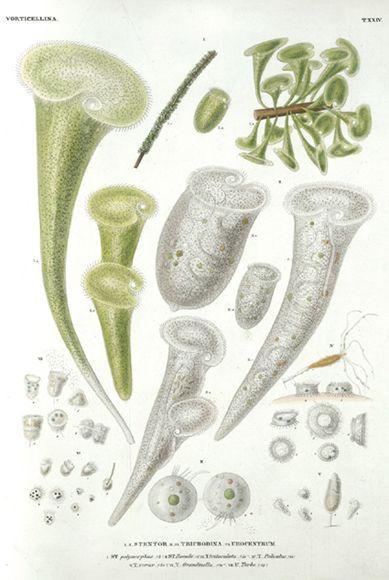
A fanfare of protozoa from a classic nineteenth-century work on microscopic organisms, Infusionsthierchen, by the German naturalist Christian Gottfried Ehrenberg. (credit 19.12)
Eventually the eukaryotes learned an even more singular trick. It took a long time—a billion years or so—but it was a good one when they mastered it. They learned to form together into complex multicellular beings. Thanks to this innovation, big, complicated, visible entities like us were possible. Planet Earth was ready to move on to its next ambitious phase.
But before we get too excited about that, it is worth remembering that the world, as we are about to see, still belongs to the very small.
![]()
1 There are actually twenty-two naturally occurring amino acids known on Earth, and more may await discovery, but only twenty of them are necessary to produce us and most other living things. The twenty-second, called pyrrolysine, was discovered in 2002 by researchers at Ohio State University and is found only in a single type of Archaean (a basic form of life that we will discuss a little further on in the story) called Methanosarcina barkeri.
The AIDS virus, HIV, greatly magnified. The virus can sit, harmless and unnoticed, in the nuclei of cells for years before springing into action. (credit 20.1)