Chapter Eight
INTRODUCTION
Even though scleractinian corals are the primary builders of present-day coral reefs, skeletal material of other groups, both plant and animal, is incorporated into the construction of the reef matrix. In fact, incorporation, cementation and consolidation of all these calcareous materials, including the corals, is a primary requirement of reef-building, since scleractinian corals rarely, if ever, do it alone. However, large aggregations of massive species, such as Pontes, can locally make a significant contribution to the reef matrix (fig. 8.1). While large aggregations of massive Pontes spp. are a common feature of many back-reef and lagoonal environments, they have also been found on the seaward slopes of actively growing reefs. In these instances, they seem to play an important role in the lateral expansion of the reefs. However, on many Indonesian coral reefs, the octocorals (Octocorallia) are a consequential, if not the dominant, component of the coral reef community in terms of abundance, biomass, as well as diversity (fig. 8.2). Class Anthozoa, to which the scleractinian reef-building corals belong, is represented by a number of other groups of organisms that deposit hard skeletons. The Octocorallia is one such group, albeit their present-day contribution to the development of reef matrix is on a "smaller" scale when compared to the scleractinian corals. From an ecological perspective, however, octocorals as well as zoanthids (Hexacorallia) are an important component of the coral reef benthic community. Octocorallia is a large, but taxonomically very poorly known, group of reef organisms, especially in Indonesian waters. So poorly are octocorals known in the archipelago, that when we asked one of the specialists of this group about a possibility of presenting a list of known octocorals from the Indonesian Archipelago, he stated that: "it is impossible to make a reliable list of Octocorallia from Indonesia" (L.P. van Ofwegen pers. comm.).
The octocorals are sedentary clonal colonial organisms. Soft corals may be viewed as opportunists, mainly because of their ability to survive in highly stressed environments, where scleractinian corals can barely manage to survive or where they have been eliminated (Sorokin 1991). For example, soft corals have been observed to monopolize entire reef habitats following major disturbances (e.g., Acanthaster planci outbreak) which caused significant mortality of scleractinian corals.
These ubiquitous reef-dwellers of Indonesian reefs have a circumglobal distribution, ranging from the frigid waters of the Arctic and Antarctic to the warm waters of shallow tropical seas. Their vertical distribution ranges from the intertidal to the abyssal, and on many Indonesian reefs they are a dominant group of organisms, especially on the deeper fore-reef slopes, where they turn the dark reef into an amazingly colourful environment, but only once the camera lights are turned on (colour plates 8.1 and 8.2).

Figure 8.1. An aggregation of Pontes sp. on a narrow reef flat, Kakaban Island (uplifted atoll). Some species may make a notable contribution to local construction of reef matrix without the assistance of cementing processes.
Photo by Tomas and Anmarie Tomascik.


Figure 8.2. Luxuriant octocoral communities dominate many reefal and non-reefal substrates in the archipelago. A) Soft-coral dominated-reef community (mostly Sarcophyton spp. and Lobophytum spp.), Lucipara Islands, Banda Sea. B) Softcoral-dominated volcanic substrate at Gunung Manuk (mostly Sinularia spp.), Banda Sea.
A. Photo courtesy of Ron and Valerie Taylor; B. Photo by Tomas and Anmarie Tomascik.
CLASSIFICATION OF SUBCLASS OCTOCORALLIA
According to Bayer (1981a), the Subclass Octocorallia is represented by three Orders: 1) Helioporacea (= Coenothecalia); 2) Alcyonacea (including the Suborders Protoalcyonaria, Stolonifera, Alcyoniina, Scleraxonia, Holaxonia [Gorgonacea = Scleraxonia + Holaxonia]); and 3) Pennatulacea. Octocorallia are sedentary anthozoans with polyps that have eight pinnate (usually) tentacles and eight unpaired mesenteries and a ventral groove on the stomodaeum (fig. 8.3) (colour plate 8.3). The majority of octocorals are firmly cemented to the substrate, but there are exceptions (e.g., Order Pennatulacea or the sea pens). The sea pens are a highly diversified group of colonial octocorals with an elongate primary polyp embedded in sediments on the sea bottom. The distal end of the primary polyp bears secondary polyps, usually on lateral branches (Barnes 1980). Sea pens date back to the Cretaceous and Tertiary. They inhabit a variety of shallow-water tropical environments from which they differentiated and dispersed into temperate regions and deep-sea environments, including the abyssal plains (Williams 1993). Highly derived taxa are present in many tropical shallow-water habitats and are sympatric with the more primitive forms (Williams 1993). Worldwide, there are about 32 genera of sea pens comprising 193 species. The Indo-Pacific has the highest species diversity of any biogeographic region, with 111 species (79 endemics) and 32 genera. The Indonesian sea-pen fauna is very poorly known. There are 12 known species of Veretillidae and four species of Echinoptilidae. According to Williams (1993), veretillid genera have the most plesiomorphic characters (i.e., primitive) of all sea pens and are concentrated in shallow-water habitats of the Indo-Pacific, whereas taxa with more apomorphic characters (i.e., evolutionarily advanced character state) are present in all seas and all depths.
The Octocorallia collected during the Siboga and Snellius Expeditions have been examined by Nutting (1911), Thomson and Dean (1931), Stiasny (1935) and Verseveldt (1960, 1966) (table 8.1). The Alcyoniidae are a very conspicuous octocoral group in terms of abundance and biomass on most Indonesian reefs. There are about 195 known species in Indonesian waters belonging to the Suborder Alcyoniina (L. P. van Ofwegen, pers. comm). The taxonomy of scleritic (i.e., sclerite-secreting) Octocorallia is based largely on sclerite morphology. The sclerites are generally embedded in the soft ectodermal and coenenchymal tissue layers. The sclerites are of different shapes and sizes, with seemingly different functional roles. The sclerites in Indo-Pacific soft corals (four species examined) comprise about 19% of total dry body weight (Cary 1931). However, Konishi (1981) reported that sclerite weight of several soft corals (alcyonaceans) from the Ryukyus and Micronesia is over 50% of dried body weight. The sclerites of the zooxanthellate genus Sinularia (fig. 8.5) are among the largest found in the group (Konishi 1981). In some species, such as those belonging to the genus Sinularia, the sclerites tend


Figure 8.3. The octocorals are characterized by eight pinnate tentacles. A) The tentacles of the organ pipe coral, Tubipora musica. B) The calcareous skeleton of Tubipora musica.
A. Photo courtesy of Ron and Valerie Taylor; B. Photo by Tomas and Anmarie Tomascik.


Figure 8.4. Octocorallia are a diverse group of reef organisms inhabiting a variety of reef habitats. A) Sea pens (Order Pennatulacea) inhabit mostly sandy and muddy habitats (Pteroeides sp.). The primary polyp is elongate with a heavily calcified central axis. B) Gorgonian fans are a common group of octocorals on Indonesian reefs. The large sea fan (centre left) is possibly Melithaea squamata.
Photos by Tomas and Anmarie Tomascik, Sangalaki Island, East Kalimantan.

Figure 8.5. The calcareous sclerites of zooxanthellate genus Sinularia. A) club sclerite; B) spindle sclerites; C) spindle sclerite; D) spindle sclerite.
Photos courtesy of LP. van Ofwegen, National Museum of Natural History, Leiden.
Table 8.1. List of Alcyoniina (families, genera and species) from the Indonesian Archipelago based on the Siboga and Snellius collections, and recent surveys conducted during the Rumphius Biohistorical Expedition to Ambon (1990).


to concentrate at the basal portion of the stalk, where they give structural support to the colony (Konishi 1981). Thus, Sinularia species are able to occupy high-energy habitats (colour plate 8.4).
REEF-BUILDING CONTRIBUTION OF OCTOCORALLIA
There are interesting exceptions to the supposition that octocorals, in general, are not important reef-builders. Some groups, such as the non-scleractinian zooxanthellate blue coral, Heliopora coerulea (Coenothecalia, Helioporidae), whose skeletal material consists of fibrocrystalline aragonite (i.e., non-scleritic aragonite), are known to form massive colonies on some Japanese reefs (Veron 1986). These corals have in fact received considerable public attention in Japan due to their rapid degradation. On some of the remote oceanic platform reefs in the Banda Sea (e.g., Lucipara Islands), H. coeruleaYias built entire reef flats. The reef flats are constructed from thousands of large H. coerulea microatolls which have joined together. The spaces between the microatolls have been long filled with reefal sediments, giving the appearance of a solid reef below. The sediments were found to be dominated by discoid benthic foraminiferans (Marginopora sp.). The inter-microatoll spaces seem to provide a favourable environment for the rare tridacnid bivalve Hippopus hippopus, which occurs in great abundance. Just seaward of the reef flat, on the shallow reef slope, large colonies of Heliopora coerulea dominate the reef (fig. 8.6). The abundance and position of these massive colonies indicate that substantial lateral reef growth may be occurring. H. coerulea contributes significant amounts of non-scleritic aragonite to the reef framework.
The contribution of other Octocorallia to the construction of the reef matrix consists of calcite sclerites (e.g., Alcyonacea, soft corals), and branched (e.g., Gorgonacea - sea fans) or unbranched (Pennatulacea, sea pens) scleroprotein skeletons (colour plate 8.5 and fig. 8.7). However, Konishi (1981) points out that even though some octocorals, such as the Family Alcyoniidae, are a major reef-dwelling soft-coral group in the Indo-Pacific, their skeletal remains (i.e., sclerites) are an insignificant (not more than 1%) component of unconsolidated reefal sediments. Nonetheless, it has been shown that alcyonacean spiculite formed almost exclusively around the basal stalks of Sinularia can be a significant local component in shallow-water high-energy habitats (fig. 8.8) (Konishi 1981). A well-cemented basal stalk is a common feature in many octocorals, such as Nephthea spp. which seem to prefer reefal habitats with strong currents and wave action.
Fossil equivalents of the alcyonacean spiculite (i.e., a sediment or rock composed primarily of alcyonacean sclerites) have been found throughout the Indo-Pacific. The alcyonacean spiculite forms through in situ submarine cementation, which protects the sclerites from dispersion and concentrates them for subsequent lithification into solid rock (Konishi 1981). The two major minerals involved in the cementation of alcyonacean spiculite are aragonite (i.e., orthorhombic CaCO3), which is the major component, and magnesium calcite (MgCO3). The large spindles of Sinularia consist mainly of aragonite, with a MgCO3 content of 12%-15%.
Some researchers are of the opinion that Alcyonacea have been greatly under estimated with regards to their reef-building capability (C. Lewis, pers. comm.). This minority view has received some new support from the results of the 1984 Snellius-II Expedition. Van der Land and Sukarno (1986) report an interesting discovery of a live, as yet undescribed, indurated alcyonacean from Kapoposang reef in the Spermonde Archipelago. During the expedition's visit to Guang Island, off the southwest tip of Selayar Island, expedition scientists found an unusual fossil that was mixed with a collection of Pliocene scleractinian corals. The fossil consisted of a lump of loosely cemented alcyonacean sclerites, which is highly unusual in itself, since cementation and fossilization of alcyonacean sclerites is accidental and a very rare phenomenon indeed. After finding additional samples from Miocene limestone deposits on Gili Lawa Laut (east of Komodo) and Sanggar Bay in Sumbawa, a conclusion was reached that the fossils must belong to an alcyonacean with a skeleton consisting of loosely cemented sclerites. An interesting question that arose was whether this alcyonacean is extant. After an organized search of the Kapoposang reef in the Spermonde Archipelago, a large brown alcyonacean colony (2 m diameter) was found whose mushroom-shaped stony skeleton was identical to the fossil samples from Guang, Gili Lawa and Sanggar Bay (van der Land and Sukarno 1986).

Figure 8.6. Octocoral-dominated reef edge of an oceanic platform reef, Lucipara Islands, Banda Sea. Blue coral (Heliopora coerulea) is a dominant component of the community.
Photo courtesy of Ron and Valerie Taylor.

Figure 8.7. The skeletons of gorgonian sea fans (2 m) consist of tough scleroprotein, Sangalaki Island, East Kalimantan.
Photo by Tomas and Anmarie Tomascik.

Figure 8.8. Schematic cross-section of Sinularia colony showing the location of the sclerites and major body parts.
From Manuputy 1989b, modified from Konishi 1981.
Octocoral-Zooxanthellae Symbiosis
Like many scleractinians, most tropical shallow-water octocorals are associated with the endosymbiotic zooxanthellae. While the scleractinian-zooxanthellae symbiosis has received considerable scientific attention, comparable research on Octocorallia is far behind, and therefore, not much is known about the functional aspects of this relationship. The nutritional role of zooxanthellae, and their possible role in the calcification process and deposition of sclerites, has not been fully investigated in soft corals, even though in scleractinians the calcification process is greatly facilitated by the association. Since the majority of Octocorallia, and many Zoantharia, are associated with zooxanthellae, their vertical distribution on the reef is restricted to the euphotic zone, where they have to compete for available space with the scleractinians (fig. 8.9). Sorokin (1991) has recently suggested that the ability of octocorals to compete with scleractinians, and other sessile reef organisms, is mainly a function of their chemical defense arsenal, and relatively fast growth rates (fig. 8.10). The Indonesian reefs are a natural laboratory, where different community interactions between soft and scleractinian corals may be studied side-by-side. It is not uncommon to view during a dive along a 200 m reef track, a scleractinian-dominated community that rapidly changes into a mixed scleractinian/ octocoral community, which in turn gives way to a soft-coral-dominated community. What biotic and abiotic factors are responsible for these community mosaics is not known.

Figure 8.9. Chemical warfare is a common occurrence on the reef. Competition for space between octocorals and scleractinians, and other benthic organisms, is intense. A) Sarcophyton and Sinularia monopolize many patches at the expense of other benthic organisms. B) Using its powerful chemical defense capabilities (i.e., highly toxic metabolites), this Sinularia sp. can grow in an optimal habitat, undisturbed by the nearby assemblage of feather stars, Oxycomanthus bennetti (Crinoidea).
Photos by Tomas and Anmarie Tomascik, Koon Island, Halmahera Sea.
Nutritional Aspects of Symbiosis. It seems that the main advantage of the symbiotic association for the soft corals is nutritional, since photosynthetic rates of octocorals range between 0.1 to 3.2 mg C.g" dry weight of colony per day, and are comparable to the scleractinians (Sorokin 1991) (table 8.2). The ratio of photosynthesis to respiration (P/R) in symbiotic octocorals studied thus far is usually greater than one, which suggests that the animals are functionally autotrophic systems (Sorokin 1991). For example, Xenia relies almost entirely on photosynthates from its zoox anthellae endosymbionts, and is a very poor heterotroph. Heterotrophic-feeding may be used purely for the supplementary uptake of nitrogen and phosphorus, which are needed for protein syntheses and energy transfer. The continuously pulsating polyps of Xenia spp. are frequently mistaken for a feeding behaviour by visitors to the reefs. The polyp contractions are purely for the intake of respiratory water (Morton 1990).


Figure 8.10. As a result of their relatively fast growth rates and potent chemical warfare arsenal, octocorals are able to colonize and monopolize new spaces when they become available. A) A large Sarcophyton colony has established itself on top of a damaged Lobophyllia hemprichii. B) A large colony of Oulophyllia under attack from encroaching soft corals (i.e., Lobophytum, Sarcophyton and Sinularia). Note the distinct white margin (dead tissue) on the Sarcophyton colony (lower left), indicating that the scleractinian is successfully defending its territory.
Sources: A. Photo by Tomas and Anmarie Tomascik; B. Photo courtesy of Ron and Valerie Taylor.
Table 8.2. Comparison of total photosynthetic (P) and respiration (R) rates in selected symbiotic octocorals and scleractinians, with corresponding photosynthesis to respiration ratio(P/R). Values for P and R in μg C.g-1 dry colony weight day-1.

Considering the external morphology of the soft coral polyps, one would assume that heterotrophic uptake of organic matter (e.g., prey capture, suspension-feeding, etc.) would be an important source of nutrients, that should satisfy a significant portion of their energy requirements. However, according to Sorokin (1991), the majority of symbiotic octocorals derive very little from heterotrophic-feeding, and, in fact, they seem to be very poor predators (table 8.3). Apparently, the octocoral eight-tentacle polyp, with fewer types of nematocysts and different tentacle morphology, is not as efficient a predator as the scleractinian polyp. According to Sorokin (1991), even asymbiotic octocorals which rely on heterotrophic uptake to satisfy their energy requirements are less efficient predators than scleractinians. Note that these studies have been conducted under laboratory conditions and focused mainly on polyp or mucus-feeding.
An interesting observation was made on Pulau Manuk, an active volcano in the Banda Sea, where soft corals are a dominant component of the reef community. Sarcophyton and Sinularia form extensive monospecific stands that support an amazing biomass of reef and reef-associated fish. Most Sarcophyton colonies were seen to feed during the day when their polyps (autozooids) were fully expanded; however, whether they were capturing zooplankton or were mainly suspension-feeding is not known (fig. 8.11). Sarcophyton and Sinularia-like colonies were up to 1.5 m, or more, in diameter, with an extensive combined surface area that seems to be an efficient filter. However, the Sinularia-like colonies were not seen with expanded polyps. During a night dive we observed numerous long, thin, translucent hair-like filaments, up to 1 m in length, trailing in the current (about 0.5 m.sec") from all Sinularia-like colonies. These feeding structures extruded from numerous pores located on the surface of the slender, finger-like lobes of the colonies and consisted of long central threads from which a number of smaller (1020 cm in length) axillary filaments branched out. The distal end of the main filament terminated in a small fan- or feather-like structure. The filaments were being actively reeled into the pores on the capitulum. Upon closer inspection, we found various suspended particulate matter attached to the filaments, which were sticky to the touch. The action of the filaments was somewhat similar to a fishing line being pulled in. The effectiveness of this feeding mechanism was revealed to us when we proceeded to take video footage. The bright lights of the camera attracted an amazing array of zooplankton that rapidly turned into a swarm. Since the lights were very close to the filaments, various zooplankton species (mysids, chaetognaths, various polychaetes, copepods, etc.) and invertebrate larvae (fig. 8.12) were seen being captured by the filaments, and immediately reeled into the pores. The polyps played no visible role in this activity, and were not expanded. The preference of Sinularia for high-energy environments (i.e., currents and wave action) may be related to its feeding behaviour, since the efficiency of the feeding mechanism observed would depend on local current conditions. Feeding experiments in the field have so far not been conducted, and the feeding mechanisms of soft corals remain poorly understood.
Table 8.3. Contribution of heterotrophic sources of organic matter to the nutritional requirements of selected symbiotic and asymbiotic octocorals, expressed as assimilation-to-respiration ratio. All values represent the percentage of respiratory losses compensated by heterotrophic uptake of organic matter through different feeding mechanisms: A) predatory feeding on zooplankton; B) feeding on algae; C) feeding on bacteria; D) uptake of dissolved organic matter (protein hydrolysate); E) total heterotrophic uptake. R Food source: rotifers; A Food source: Artemia nauplii.


Figure 8.11. Sarcophyton sp. with fully expanded polyps on the capitulum. Note small pores on the surface of the capitulum.
Photo by Tomas and Anmarie Tomascik.
The Propagation of Symbiosis. Recent studies suggest that the symbiotic relationship between octocorals and the zooxanthellae is renewed each generation by sexually produced offspring (Benayahu 1993). However, eggs of broadcast-spawners, such as Cladiella, Sarcophyton and Sinularia, and surface-brooders such as Clavularia and Parerythropodium, do not contain zooxanthellae upon spawning, and the juveniles must acquire zooxanthellae from ambient seawater (Benayahu 1993). Other soft corals such as Heteroxenia spp. release non-zooxanthellate planulae. Unlike the majority of scleractinians, whose planulae settle with a full complement of zooxanthellae, zooxanthellae in the octocorals are recruited into the newly settled polyps after metamorphosis has been completed. However, in some brooding genera, such as Xenia and Sympodium, zooxanthellae are acquired before the planulae are released, as is often the case with scleractinian brooding corals (Tomascik and Sander 1987b).


Figure 8.12. In addition to the holoplankton that is continuously being pumped into the reefal systems from the surrounding ocean, the reefal waters are also rich in planktonic larval stages of its benthic inhabitants. A) A zoea stage of a reef-dwelling crab (Brachyura). Brachyuran larvae, or zoea larvae, are easily recognizable by their rounded carapace, which is ornamented with prominent spines that may serve as antipredatory defenses, or aid in flotation. B) Shrimp larvae are another important component of the planktonic community (Panaeidae; Gennadas sp.)
Photos courtesy of R. Steene.
The process by which the zooxanthellae infect the planula before it is released has been recently investigated in the brooding octocoral Xenia macrospiculata. In this group of gonochoric (i.e., dioecious - each colony being of different sex) symbiotic octocorals, fertilization of azooxanthellate eggs is internal (i.e., inside the polyp), and the planulae develop in specialized brooding pouches located near the anthocodial bases (Achituv et al. 1992). Embryogenesis proceeds within these pouches, and the planulae containing zooxanthellae are shed through temporary openings between the polyps (Achituv et al. 1992). It appears that the planulae acquire the zooxanthellae by means of endocytosis by ectodermal amoeboid cells which phagocitize the zooxanthellae at the outer surface of the planulae. The ameboids then traverse through the planulae tissue and penetrate the endoderm where the zooxanthellae are deposited. According to Achituv et al. (1992) the origin of the zooxanthellae is most likely the endoderm of the brooding pouches.
Reproduction. As with the scleractinians, there is little data available on the life history strategies and reproductive biology of Octocorallia in the Indonesian Archipelago. In fact, a similar situation exists elsewhere, even though some progress in this field has recently been made (Benayahu and Loya 1983, 1984a, 1984, 1986; Achituv et al. 1992; Benayahu 1993). Octocorals rely on both asexual and sexual modes of reproduction. The most common type of asexual reproduction in alcyonacean corals is through fragmentation (Walker and Bull 1983), or by vegetative propagation as in some nephtheids and xeniids (Tursch and Tursch 1983). The most studied soft corals are the Xeniidae (e.g., Xenia macrospiculata, Heteroxenia fuscescens), a group of gonochoric-brooders, which release planulae containing zooxanthellae. Benayahu and Loya (1985) have discovered that in Xenia macrospiculata asexual reproduction is accomplished by colony fission, a previously unknown mode of asexual reproduction in alcyonaceans.
Lobophytum crassum is among the very few alcyonaceans whose reproductive behaviour has been studied (Yamazoto et al. 1981). It is one of the most common soft corals on Indonesian reefs, yet nothing is known about its life history. As with the other alcyonaceans, L. crassum is a gonochoric species, with gonads being developed on autozooid mesenteries. Unlike the xeniids, however, L. crassum appears to be a broadcast-spawner, with external fertilization. In fact, Babcock et al. (1986) reported that seven alcyonaceans (Alcyoniidae) were observed to spawn (i.e., broadcast-spawners) during die mass-spawning event in 1983 on the Great Barrier Reef. The species involved were Alcyonium aspiculatum, Lobophytum compactum, L. microlobulatum, L. hirsutum, Sarcophyton sp., Sinularia c.f. deformis, and S. polydactyla (Babcock et al. 1986).
Another unique feature in the reproduction of L. crassum is prolonged oogenesis. Oocyte maturation requires 22-23 months, which is very similar to Sarcophyton glaucum (Benayahu and Loya 1986). S. glaucum is also a gonochoric broadcast-spawner, with a single annual spawning event that appears to last only one night. The characteristic feature of this group of octocorals is their large body size and long life spans. Their reproductive effort is high, compared to many other reef organisms, yet very little is known about the evolutionary significance of this mode of reproduction.
The reproductive strategy of Parerythropodium fulvum fulvum, an encrusting form, is somewhat different from die otiier alcyonaceans. As widi most alcyonaceans studied, P.f. fulvum is gonochoric with annual spawning events, which are synchronized by lunar periodicity (Benayahu and Loya 1983). Fertilization is internal, and while the species is fully oviparous (i.e., egg-laying) the planulae are brooded on the surface of female colonies, entangled in a mucous suspension (Benayahu and Loya 1983). This type of "parental care" is of considerable adaptive value for species whose fecundity (i.e., number of eggs produced) is limited by their body size (i.e., thin coenenchyme and small polyps). Benayahu and Loya (1983) suggested that this type of reproduction is an adaptation of encrusting species to maximize their reproductive success (i.e., number of recruits). Thus, low fecundity (e.g., 21 eggs per polyp) is compensated for by "brooding protection" which may substantially increase the survivorship of planulae to the settlement stage.
General Distribution. In general, the azooxanthellate octocorals, especially the gorgoneans, tend to dominate the deeper parts of the reef, while the zooxanthellate groups (e.g., Sinularia, Lobophytum) dominate in the shallower habitats. However, some zooxanthellate species, such as Sinularia dura, can be the dominant component of deep reef slopes.

Figure 8.13. A shallow, subtidal seaward edge of an oceanic platform reef dominated by an octocoral community, Sarco phyton sp. and Heliopora coerulea.
Photo courtesy of Ron and Valerie Taylor.
The dominant alcyonarean genera in high-energy environments are Lobophytum, Sarcophyton and Sinularia (fig. 8.13). Their ability to survive and flourish in high-energy environments is related to their strong basal attachment to the substrate, and their relatively flexible body column, which can withstand strong current velocities and wave action. Indeed, in some areas where currents are exceptionally fast (e.g., Batu Kapal, Banda Islands), soft corals, Sarcophyton, Lobophytum and Sinularia as well as Tubastraea micrantha, are the dominant anthozoans. T. micrantha is an azooxanthellate scleractinian, which is frequently very abundant on high-energy fringing reefs, such as those along the south coast of Java, where it may be considered as a reef-builder in a number of localities (T. Tomascik, pers. obs.).
In many back-reef areas, or in sheltered, more turbid lagoonal environments, Sarcophyton can also be found to dominate the reef community. Alcyonarian-dominated reef communities in Indonesia seem to be especially common in either coastal turbid waters or on oceanic reefs subjected to strong upwelling. In a study of soft coral distributions in Kepulauan Seribu, Manuputty (1992) reported that there was a strong relationship between percent cover by Sinularia polydactyla and the distance from shore (i.e., from Jakarta). However, reexamination of the data revealed that no significant correlation exists between either the abundance or percent cover by soft corals, and water transparency. A major difficulty in analyzing the data is that the environmental data set (Secchi disk values) was compiled in 1985. Clearly, more in-depth ecological and environmental studies are needed to answer these questions. Nonetheless, a similar pattern was observed in the Berau Islands in East Kalimantan. The water quality of the nearshore reef environments (i.e., fringing reefs), north of Tanjung Batu, can be classified as turbid, yet the reefs are dominated by alcyonacean communities. In fact, on many of the inshore reefs, where Secchi disc visibility is less than 3 m, and where underwater horizontal visibility is 2 m or less, the soft corals (Sarcophyton and Sinularia) are the dominant group in terms of their abundance and percentage of substrate cover, which can be as high as 80% -100%. About 15 km offshore, towards Darawan Island, the shelf of East Kalimantan is dotted with numerous shelf reefs of considerable size (up to 150 km ). Many of these reef platforms are separated from one another by deep (>30 m) and relatively narrow channels (< 500 m) through which tidal currents flow with velocities in excess of 4 m.sec". The reef slopes in many of the channels are dominated by soft coral communities. Furthermore, some of the large platform reefs (e.g., Karang Batu or Pulau Panjang Reef about 10 km offshore) are cut by numerous reef channels that are between 10-20 m wide, and 5-10 m deep. These were discovered on-site, since none of the available hydrographic charts are on scales large enough to map these features. The bottoms of the channels are pure carbonate sand, even though the reefs are located in close proximity to a major river (i.e., the Berau River). It seems that most of the suspended material carried by the river is deposited within about 15 km of the delta, and only fine particulate matter is transported over longer distances offshore, mainly as a result of strong currents. At two locations, the reef slopes in the channels were dominated by soft coral communities, with only a few scleractinians present. These communities extended for hundreds of metres (surveyed by Manta tows), and were breached either by mixed scleractinian-octocoral assemblages or by 10-20-m-wide tide channels consisting of sand and rubble. Because of the high tidal range (i.e., about 3 m), tidal currents through the reef channels are exceptionally fast. Interestingly enough, the bottoms of the reef channels are important habitats of Tridacna gigas. During the Manta tows, we observed numerous individuals that were in excess of 1 m shell length. In addition, large Petrosia testudinaria (1.5 m high and 2 m diameter) were very common.
Thus, we have observed reefs and reef zones dominated by Sarcophyton and Sinularia in a variety of environments, ranging from the inshore fringing reefs of East Kalimantan, where they lie adjacent to tropical rain forests, to the oceanic platform reefs of the Banda Sea, where they are hundreds of kilometres from the nearest land.
Chemical Warfare on the Reef. The great abundance of Octocorallia on many Indonesian reefs may be directly related to their ability to produce potent natural compounds (i.e., terpenoids), which have a number of ecological functions. In a study on defensive strategies of soft corals, La Barre et al. (1986) demonstrated that 50% of soft coral extracts tested were strongly ichthyotoxic, and 88% were considered as feeding deterrents. Perhaps more significant was the finding that feeding deterrence was also common among non-toxic soft corals. The most important role of secondary metabolites in soft corals seems to be anti-predatory defense (i.e., ichthyotoxicity) (Bakus 1981; Coll et al. 1982a, b; Coll and Sammarco 1983; Alino et al. 1988). The metabolites are very effective against many potential reef predators, and were shown to be potentially lethal to the test fish Gambusia affinis (Coll et al. 1982a; Coll and Sammarco 1983).
Evolution of anti-predatory defenses through morphological, chemical andbehavioural adaptations is a common phenomenon in nature (La Barre et al. 1986), and nowhere are these defensive mechanisms more diverse than in the tropical ecosystems. The two main anti-predatory defenses in Octocorallia function as feeding deterrents through direct toxicity which results in severe stress or mortality of the predator, or more commonly, they work at the olfactory and taste sensory levels. The secondary metabolites are either synthesized directly by the prey, or they are acquired by the prey from its main food source (La Barre et al. 1986). The major metabolite in Sinularia maxima, for example, is a cembranoid diterpene (fig. 8.14), which is highly effective against most predators at normal concentrations, and is synthesized by the soft coral. Under experimentally produced high concentrations, the diterpene also becomes a deterrent to one of its major predators, the butterflyfish Chaetodon unimaculatus. Apart from the few predators that have found ways to deal with the toxic metabolites, soft corals are practically immune to predation (Bakus 1981). In addition to the toxins that are quite potent, the coenenchyme of many octocorals contains an abundance of calcareous sclerites (sharp sclerites), which, in addition to their structural function, provide considerable protection from predation. Alino et al. (1988) have found that crude extracts from Sinularia flexibilis were highly toxic towards Gambusia affinis. However, some soft coral predators, such as the egg cowry, Ovula ovum (fig. 8.15), the teardrop butterflyfish, Chaetodon unimaculatus (colour plate 8.6) or the black damselfish Neoglyphidodon melas, and the nudibranch Phyllodesmium longicirra, most likely possess specialized physiological adaptations (e.g., production of antitoxins) to neutralize the toxins. Wylie and Paul (1989) have shown that Chaetodon unimaculatus is immune to the toxicity of the metabolites which pass through the digestive system of the butterflyfish without any breakdown or neutralization into other less toxic compounds. In contrast, Coll et al. (1983) reported that the egg cowry, Ovula ovum, which feeds almost exclusively on Sarcophyton spp., is able to convert the toxic terpene into a harmless compound.
Many of the natural terpentine compounds, present in most soft corals, may turn out to be economically important (e.g., pharmaceutical or industrial) bioactive substances. Considering the abundance of Octocorallia, and their obvious economic potential, it is, therefore, very surprising that the group is one of the least studied and understood of reef organisms in the Indonesian waters (Manuputty 1992). Octocorallia are, however, an extremely difficult taxonomic group (i.e., at species level), exhibiting great intraspecific variability of form, which may partly explain why they have been largely neglected (van Ofwegen 1987) since the time of the major expeditions (i.e., Siboga, Snellius). With the exception of some rudimentary ecological surveys (Manuputty 1990a,b; 1992), nothing is known about their distribution within the archipelago (see box 8.1; van Ofwegen).
Recently some progress has been made, as for example, at Diponegoro University (UNDIP), Semarang, where preliminary research shows great promise that some soft coral species contain natural compounds with powerful antifouling properties (M.J. Risk, pers. comm.). The UNDIP - McMaster University, Canada, joint marine research project is an excellent example where collaborative research efforts between Indonesian and foreign universities have produced excellent results. However, taxonomic research is lagging far behind, and considerable efforts should be made to develop new working relationships with foreign institutions where training of Indonesian taxonomists may take place, with the National

Figure 8.14. Chemical structure of cembranoid diterpene isolated from Sinularia maxima.
Source: Wylie and Paul 1989.

Figure 8.15. Egg cowry, Ovula ovum, feeding on Sinularia sp. in Pulau Panjang, Berau Islands, East Kalimantan.
Photo by Tomas and Anmarie Tomascik.
Box 8.1. The present status of taxonomic knowledge of octocorals in Indonesia.
LP. van Ofwegen, National Museum of Natural History, Leiden, the Netherlands.
Bayer (1981b) reviewed the status of taxonomic knowledge of octocorals in the major geographical regions of the world, and recognized four categories. He placed the central part of the Malay Archipelago in his second category of "Moderately well-known: where there is extensive literature, but many more species remain to be described and taxonomic problems to be solved, and the major patterns of distribution must yet be worked out".
The rest of the Malay Archipelago was placed in his third category of "Poorly known: where the literature is sparse and incomplete. Here the major part of the fauna remains to be described and a large number of species will inevitably be new to science. The major faunal relationships can be deduced, but distributional patterns are not clearly understood", and the fourth category of "Minimally known: where the literature consists of scattered taxonomic descriptions and isolated distributional records, many of them inadequately substantiated. Most of the basic descriptive work remains to be done".
Central Indonesia deserved its place in the second category because of the extensive collection made by the Siboga Expedition, almost one hundred years ago. Bearing in mind that this collection was mainly obtained by dredging, and contained almost no specimens from shallow water, I consider the reefs of central Indonesia to be in Bayer's third category "poorly known". For example, the soft coral Family Alcyoniidae, mainly occurring in shallow-water habitats, had until 1994 only 12 species described from Ambon. After identifying a small collection of the 'Rumphius Biohistorical Expedition (1990)' the number of species rose to 44 (van Ofwegen and Vennam 1994). Undoubtedly, many more species will be found after extensive collecting. Therefore, the "moderately well-known" categorization of central Indonesia only applies to the deep-water octocorals.
Further information on Indonesian soft corals is mainly based on the first Snellius Expedition conducted 65 years ago. Although more species have been described from Indonesia (see Bayer, 1981b: 31), the vast majority of species come from the two above-mentioned expeditions.
In 1981, the octocorals from Indonesia were considered poorly known. What has changed since then? Not much, I have to conclude. Verseveldt (1983) described Lobophytum hapalolobatum (Alcyoniidae) from the Java Sea; Verseveldt and Bayer (1988) described Eleutherobia sumbawaensis (Alcyoniidae) from Siboga material; I described Wrightella stiasnyi (Melithaeidae) from the Snellius Expedition material (van Ofwegen, 1989) and Sinularia slieringsi (Alcyoniidae) from Ambon (van Ofwegen and Vennam 1994). In 15 years only four new species were described, quite a discrepancy with Bayer's earlier statement that "many more species remain to be described".
Why is there no progress in octocoral taxonomy in Indonesia? First of all the taxonomy of octocorals is still in a very incomplete state. Many revisions are needed before species can be identified with any certainty. However, since the revisions of the three most common genera of the Family Alcyoniidae, namely Sinularia (Verseveldt 1980), Sarcophyton (Verseveldt 1982) and Lobophytum (Verseveldt 1983), species identification of these genera is more straightforward. Nevertheless, very little progress has been made concerning taxonomic work in these genera. Apparently, even when revisions are available, nothing seems to happen. Therefore, the main reasons for the lack of any significant progress in Indonesia must be found elsewhere. Certainly, the lack of specialists is one of the reasons. Less than 10 specialists worldwide are working on the taxonomy of octocorals, some of them only on a part-time basis. One would expect a huge demand for identifying octocorals from Indonesia coming to these few specialists. In practice there is hardly any demand at all. Therefore, it seems that the main reason for lack of progress in octocoral taxonomy in Indonesia is a total disregard of octocorals as key components of coral reef ecosystems. Probably most people are not even aware that there are such animals as octocorals, since most of the information received by the public is mainly about the stony corals.
Considering that octocorals are, after the stony corals, the second-most common component on many coral reefs in the archipelago, the present lack of interest in this group of reef organisms (by the Indonesian scientific community) is totally unjustifiable. Even though coral reef conservation and management efforts in Indonesia have gained considerable momentum, and government recognition, octocorals are still being ignored, which is a cause for concern. It is clear that the potential economic benefits that may flow from major research efforts of this fascinating group of coral reef organisms (e.g., biotechnology) have not been fully evaluated either by the Indonesian scientific body or by government agencies. As this situation can only change by increasing the awareness of the people as well as policy and decision-makers, I am pleased that I have this opportunity to at least draw some attention to the significant lack of taxonomic knowledge of octocorals in the Indonesian Archipelago.
References
Bayer, F.M. (1981b). Status of knowledge of octocorals of world seas. Seminarios de Biologia Marinha: 3-11, bibliography: 29-102. Academia Brasileira de Ciencias, Rio de Janeiro.
van Ofwegen, LP. (1989). On Wrightella coccinea (Ellis & Solander, 1786) and Wrightella stasnyi spec. nov. (Anthozoa: Gorgonacea: Melithaeidae). Zool. Med. Leiden 63: 27-43.
van Ofwegen, LP. and Vennam, J. (1994). Results of the Rumphius Biohistorical Expedition to Ambon (1990). Part 3. The Alcyoniidae (Octocorallia: Alcyonacea). Zool. Med. Leiden 68: 135-158.
Verseveldt, J. (1980). A revision of the genus Sinularia May (Octocorallia, Alcyonacea). Zool. Verh. Leiden 179: 1-128.
Verseveldt, J. (1982). A revision of the genus Sarcophyton Lesson (Octocorallia, Alcyonacea). Zool. Verh. Leiden 192: 1-91.
Verseveldt, J. (1983). A revision of the genus Lobophytum Von Marenzeller (Octocorallia, Alcyonacea). Zool. Verh. Leiden 200: 1-103.
Versveldt, J. and Bayer, F.M. (1988). Revision of the genera Bellonella, Eleutherobia, Nidalia and Nidaliopsis (Octocorallia: Alcyoniidae and Nidaliidae), with descriptions of two new genera. Zool. Verh. Leiden 245: i-v, 1-131.
Museum of Natural History in Leiden as an important possibility.
CLASS HYDROZOA
Introduction
The fire and rose corals, although generally referred to as corals, do not belong in the Anthozoa but in the Hydrozoa. The Hydrozoa represents a group of cnidarians with a characteristic 'alternation of generations' between sessile hydroid and planktonic medusoid forms (Mather and Bennett 1993). Hydrozoan polyps, while exhibiting the general body plan of the cnidarians, lack mesenterial partitions, and their endoderm lacks nematocysts.
Order Milleporina
"Fire" corals (Milleporidae) belong to the Order Milleporina, a group of colonial hydrozoans that produce sometimes massive, but brittle, calcareous skeletons (Veron 1986). The genus Millepora is found on reefs throughout the tropical seas, from the Indo-Pacific to the Caribbean, where it inflicts painful burning stings to the unwary visitor with its minute multiple club tentacles. The only "corals" in the Class Hydrozoa are represented by the Family Milleporidae.
While not a species-rich genus, Millepora shows remarkable phenotypic plasticity wherever it is found. Fire corals can be foliaceous, massive, branching or encrusting. In the field Millepora may at first glance be confused with Heliopora coerulea, which it superficially resembles in general morphology. Closer inspection will however reveal that the surface of the coral is covered by numerous small pores without septa, called dactylopores and gastropores. Each dactylopore houses a fine hair-like dactylozooid which expands and retracts like other polyps. Each dactylozooid bears a number of mouthless polyps that are used for prey capture and defense. Food is ingested by gastrozooids which are housed in slightly larger gastropores which are dispersed among the dactylopores (fig. 8.16).
Hickson (1898a,b, 1899) was first to seek an explanation for the remarkable variation of growth-forms exhibited by this group of reef-builders, and suggested that this was caused mainly by local environmental factors. He concluded that all growth-forms were a manifestation of a single species, Millepora alcicornis L., a well-known Caribbean species. Hickson's (1899) conclusions were based on observations made on the extreme variation in a number of Millepora specimens from North Sulawesi; however, he overlooked the possibility that the growth-form might be a manifestation of a particular species in the genus. Unfortunately, he did not provide environmental notes in the reports.
The Millepora "problem" attracted the attention of H. Boschma, who from 1920 to 1922 conducted research at Edam Island (Damar Besar) in Jakarta Bay. Based on his field observations, having found a number of Millepora colonies of distinctly different morphologies side-by-side, yet exposed to the same environmental conditions, Boschma (1948) concluded that environmental conditions alone could not account for the morphological differences as suggested by Hickson (1899). This conclusion was partially supported by the fact that in a number of instances where adjacent Millepora colonies of different morphologies were close enough to be touching, they did not make clean contacts, as would be expected if they were of the same species. Boschma found that the slender species Millepora murrayi was overgrown by a more robust species, Millepora platyphylla. However, this did not eliminate the possibility that the morphology of Millepora was not susceptible to environmental influences, although, to a lesser degree than was originally envisaged by Hickson (1899). Boschma (1948) recognized 10 species "groups" in the genus Millepora, some groups with two subspecies or species. The genus Millepora in the Indonesian waters is represented by eight species, namely Millepora dichotoma, M. exaesa, M. intricata, M. latifolia, M. platyphylla, M. tenella, M. murrayi and M. tuberosa. However, since the work of Boschma, research on this group of important non-scleractinian reef-builders in Indonesia has been greatly neglected.

Figure 8.16. Diagram of Millepora polyps.
From Mapstone 1990.
Order Stylasterina
The delicate "rose" corals belong to Order Stylasterina (colour plates 8.7, 8.8). Moseley (1880) placed the Families Milleporidae and Stylasteridae in the Hydrocorallinae even though they had very little in common. However, Boschma (1951) did not regard Hydrocorallinae as a systematic unit. In his treatment of this particular group, Hickson (1906) regarded the Order Milleporina as primitive Hydrozoa, while considering Stylasterina to be a specialized group. Hyman (1940) also classified the two groups in different orders, namely Milleporina and Stylasterina, basing her decision on the morphology of the soft parts of the organisms.
Stylasterina is a large order, with distribution from the Arctic through the tropics to the Antarctic, where they occupy a variety of habitats ranging from shallow, subtidal environments to depths greater than 500 m. Unlike the Order Milleporina, Stylasterina do not have free medusae, and gametes are formed on aborted medusae in sporosacs, which are specialized cavities. At least 88 species and subspecies have been subscribed to the genus Stylaster, 25 species to Distichopora, 24 to Errina and 14 to Crypthelia (Boschma 1951, 1957).
The genus Stylaster is mainly cryptozoic, and represented by eight relatively common species throughout the Indonesian Archipelago. The species in this group are always arborescent, with very fine and delicate branches. Their characteristic feature is the presence of cyclosystems, which are groups of dactylozooids and gastrozooids. The most common shallow-water species is Stylaster elegans, which prefers cryptic habitats with strong currents and is found throughout the archipelago. Stylaster amphiheloides is very common in the eastern regions of the archipelago (e.g., Halmahera to Seram), and bears great similarity to specimens described from the Cape of Good Hope (Boschma 1957). Other relatively common species are Stylaster bellus (e.g., Sulu, Waigeo; Halmahera), Stylaster bilobatus (e.g., Waigeo, Kai Islands, Timor) and Stylaster multiplex (e.g., Sulu Islands, Seram). Some deep-water species found in the archipelago have a worldwide distribution. For example, Stylaster densicaulis found in Waigeo and Seram is similar to specimens described from Rio de la Plata, while Stylaster duchassaingi collected off Halmahera and the Sangir Islands has been described in the West Indies off Brazil as well as in the Okhotsk Sea. Stylaster gemmascens collected off the Sulu Islands has also been described from the Izu Peninsula, Japan.
While Stylaster is mainly a cryptozoic genus, Distichopora is represented by both cryptic species, which occupy similar habitats as Stylaster, as well as species living fully exposed in a number of habitats, including high-energy environments (Tomascik and Mah, pers. obs.). Distichopora spp. have mainly arborescent forms with somewhat flattened and blunt-ended branches (Veron 1986). This genus differs from Stylasterhy the absence of cyclosystems. In Distichopora, the gastropores are on the lateral margins of branches with rows of dactylopores on either side (Veron 1986). Distichopora seems to be especially abundant in the Sangihe Archipelago, North Sulawesi, where deep-blue to purple colour-types predominate. The genus Distichopora is represented by 26 species, three of which are very common in the shallow-water habitats throughout the archipelago. Distichopora coccina is common, especially in the eastern regions (e.g., Seram and Banda Islands). Rumphius may have been the first to collect this species in Ambon. Distichoporafisheri is more common in the western regions of the archipelago (e.g., Pulau Pasu; West Sumatra), while Distichopora violacea is very common in the northern and southern regions of the archipelago (e.g., North Sulawesi and Timor).
Genus Stenohelia is represented by eight species, four of which are found in Indonesia (Boschma 1957). Stenohelia minima has been found in the waters off Halmahera, and has a wide distribution. It has been described from Mauritius (Boschma 1957). Stenohelia tiliata described from specimens collected in the Sulu Islands is also found in the Cape Verde Islands in the Atlantic, while Stenohelia virgins found in Waigeo, Irian Java, has been described in the West Indies (Boschma 1957). Stenohelia umbonata collected in North Sulawesi has so far not been found in other regions, and may be endemic to this region.
Genus Crypthelia is represented by 13 species worldwide, six of which are found in the Indonesian seas. However, since the Siboga Expedition, not much has been added to the meager collection from Indonesia. The species recorded from Indonesia are Crypthelia balia (North Seram), C. pachypoma (Halmahera), C. platypoma (Waigeo), C. pudica (Moluccas), C. ramosa (North Sulawesi) and C. stenopoma (Seram, Lucipara Islands; Banda Sea). C. stenopoma has a wide distribution in the Indo-Pacific, from the Galapagos (east Pacific) to the Maldives Islands (central Indian Ocean). C. pachypoma is also found in the Galapagos Islands.
Genus Conoporais represented by five species, one of which (i.e., C. major) has been found in the Kai Islands and North Sulawesi. Genus Errina with 27 species is poorly represented in Indonesia, with only two species, namely Errina horrida found in Halmahera, and Errina ramosa in Timor. However, the monospecific genus Steganopora seems to be endemic to Indonesia, with Steganopora spinosa found in Halmahera. There are eight additional genera without any species representation in the Indonesian waters (i.e., Astya, Errinopora, Paraerina, Errinopsis, Phalagopora, Congregopora, Pliobothrus, Sporadopora).
THE NON-SKELETAL ANTHOZOA
Introduction
The Indonesian sea anemones (Anthozoa) are taxonomically as neglected as the Octocorallia, even though both groups are highly visible components of the coral reef community. To date, very little taxonomic, biological or ecological research has been conducted in Indonesia on this fascinating group of benthic reef invertebrates. Their treatment following the Octocorallia and Hydrozoa is because, unlike the former groups, sea anemones are non-skeletal reef organisms which do not contribute to the formation of the reef matrix.
Sea anemones are found throughout the world, but they are most abundant and diverse in the tropical regions, especially in shallow-water habitats such as coral reefs. They are found in a wide variety of habitats, from muddy lagoonal environments to exposed shallow-water reef crests. They vary in size from only a few millimeters tall to giant forms up to 1 m in diameter. One of their most interesting features is that many are symbiotic with photosynthetic endosymbionts, as well as with a variety of organisms ranging from small crustaceans to fish (colour plate 8.9).
Our current knowledge of this group of fascinating, but poorly known, reef-dwellers in Indonesian waters is succinctly summarized in the following section (box 8.2) by J. C. den Hartog, a leading authority on the subject.
Box 8.2. The sea anemone fauna of Indonesian coral reefs.
J.C. den Hartog, National Museum of Natural History, Leiden, the Netherlands.
INTRODUCTION
Sea anemones is a collective name for various orders of soft-bodied, non-skeletal Anthozoa comprising the Actinaria (sea anemones sensu stricto), Ceriantharia (tube anemones), Zoantharia (encrusting or colonial anemones), Corallimorpharia (coral anemones or false corals) and the very small group of the Ptychodactyaria. Together with the skeleton-forming Scleractinia (stony corals) and Anthipatharia (black corals), these groups of organisms are united in the Subclass Hexacorallia. Presumably because the old established name "Hexacorallia" is not strictly appropriate (i.e., the organization of the hexacorallian polyp is by no means always hexamerous), the name "Zoantharia" is increasingly used for the subclass, especially in the Anglo-American literature, and the names Zoanthidea or Zoanthinaria for the order. Currently, these latter names are also being used in the "Zoological Record", a catalogue of zoological literature published yearly since 1865 by the Zoological Society of London, and in general use, especially by taxonomists, throughout the world.
Thus far, the sea anemone fauna of the Indonesian Archipelago has not been the subject of extensive taxonomic investigations. The studies that are available are largely concerned with the fauna of Amboina (i.e., Ambon) and Ternate (Kwietniewski 1896, 1897, 1897a; Roule 1909). However, additional information occurs scattered in the literature. Studies from adjacent regions, such as Torres Straits (Haddon and Shackleton 1891; Haddon 1898) and the Great Barrier Reef (Saville-Kent 1893; Carlgren 1937, 1950; Muirhead and Ryland 1993), bear considerable relevance to Indonesian waters. Since many tropical Indo-Pacific species have a wide distribution, the classic studies on Red Sea and east African anemones (Klunzinger 1877; Carlgren 1900), as well as a recent paper by England (1987) on Actinaria from the Red Sea and the Indo-Pacific, bear also relevance to Indonesia.
It may be something of a shock to learn that the sea anemones collected during the famous Dutch Siboga Expedition (1899-1900), which perhaps should have formed the basis for sea anemone studies in Indonesia, were mislaid. They were intended to be studied by McMurrich (University of Toronto, Canada), but except for a monograph on the Ceriantharia (McMurrich 1910) the work was never completed. After McMurrich's death, the material (the Ceriantharia included) was never returned to its depository, the Zoological Museum in Amsterdam. Later on, when action was taken, the collections and any possible manuscripts proved untraceable; a single box with histological slides (now in Amsterdam) was all that could be secured. It seems very doubtful that any of it will ever be recovered. Fortunately, new large collections of Indonesian sea anemones were made since 1984, during Indonesian-Dutch expeditions in the Archipelago, viz. the Snellius II Expedition (1984), the Rumphius Biohistorical Expedition to Ambon (1990) (cf. Strack 1993) and an expedition to Lembeh Strait, North Sulawesi (1994). The majority of the samples of these expeditions were deposited in the National Museum of Natural History at Leiden, and are now being studied or are available for study.
THE SEA ANEMONES
Order Ptychodactyaria
Ptychodactyaria is a little-known, very small, yet spectacular group of highly specialized, mobile cold-water anemones with bipolar distribution, possibly all feeding on octocorals (cf. e.g., England and Robson 1984; Dayton et al. in press). Only three species have been described to date. The order is not likely to be represented in Indonesian waters.
Order Ceriantharia
Ceriantharia (colour plate 8.10) is a small, conspicuous group of remarkable sea anemones, presumably comprising not more than some 50-100 species worldwide. Unfortunately, not a single taxonomist is presently seriously occupied with the group.
The number of known larval forms, usually described under different names, is about twice as large as that of the adult forms. The life cycle of only a few species is known (Riemann-Zürneck 1969), but in most cases it is not established which larval and adult forms belong to the same species, which hinders a uniform classification. Since the taxonomy of the group is based primarily on internal anatomical and nematocyst characters, it is generally impossible to even distinguish genera, let alone species, without dissection and the aid of a microscope. As a result, describing and identifying Ceriantharia is currently a very time-consuming task.
The vertical distribution of Ceriantharia ranges from intertidal pools and muddy flats down to the deep sea. On Indonesian reefs and in other shallow-water habitats, cerianthids may be quite common on muddy and sandy patches as well as in mud-filled crevices among dead coral. It is likely that the number of species on Indonesian reefs does not exceed 10. Unfortunately, all published records of Indonesian species were imperfectly described. Kwietniewski (1897a) described three new species of Cerianthus on the basis of external characters alone. McMurrich (1910), though hesitatingly, identified part of the Siboga shallow-water material with Kwietniewski's species. In addition, he described one new species, Pachycerianthus monostichus, and four new larval forms. Roule (1909) identified specimens from Ambon with the east African Cerianthus maua, though on very vague grounds. Lastly, Carlgren (1924) described two species from the Kai Islands, Pachycerianthus plicatus and Isarachnanthus bandanensis. Unfortunately none of the authors provided substantial data on the cnidom. A major revision of the group is most desirable. With one or two exceptions, it is virtually impossible to accurately identify Indonesian species on the basis of available descriptions.
Ceriantharia live in self-produced flexible tubes, which in some species may reach up to 1 m in length. These tubes are not formed by mucus and foreign material as is the popular belief. Ceriantharian tubes consist predominantly of the long tubes of special atrichous nematocysts (generally referred to as ptychocysts), which are massively secreted by the body ectoderm, forming dense and very tough mats (Mariscal et al. 1977; den Hartog 1977) (fig. 8.17).
Throughout their distributional range, including Indonesian reefs and other shallow-water habitats, cerianthids are commonly associated with horseshoe worms, Phylum Phoronida (presumably the cosmopolitan Phoronis australis) (see Emig 1979). The sessile suspension-feeding phoronids are generally located under the surface layers of the smooth inside of the upper part of the cerianthid tube, only their respiratory and filter-feeding apparatus (lophophore) emerging. A single cerianthid tube may be infested by dozens of longitudinally arranged phoronids.
By day many cerianthids live fully retracted in their tubes, but in some species the unexpanded to semi-expanded double tentacle crown (divided in so-called oral and marginal tentacles) may be seen to extend slightly from the end of the tube. Most species, however, are nocturnally active and only expand after or just before sunset. To my knowledge there are no zooxanthellate species of Ceriantharia. Small fishes may be seen hovering among the tentacles (fig. 8.18) and small bivalve molluscs (Family Montecutonidae) may be encountered just under the surface of the soft substratum surrounding the tube (cf. e.g., Morton 1989).
Some crabs also associate with cerianthids, a beautiful example being the pied, white and brown Lissocarcinus laevis, which, when disturbed, quickly enters the tube of its host to hide. Shrimps of the genus Periclimenes, such as P. magnificus, which generally associate with Actinaria, may occasionally also be found on Ceriantharia and the same holds for Thor amboinensis (colour plate 8.11), which loosely associates with many Anthozoa, including stony corals.
Order Actinaria
The Actinaria form the largest and the most diverse order of sea anemones. They range from the upper littoral zone to the deepest ocean trenches (Carlgren 1956). Considering that many species described under different names from-different geographical areas are undoubtedly conspecific, and that many new species are still to be discovered, the number of species worldwide may amount to about 800-1000. In spite of large gaps, our knowledge of the group as a whole is better than that of the other orders of sea anemones. Nonetheless, up-to-date monographs of families and major genera are completely wanting, perhaps with the exception of the revision of the clownfish anemones by Dunn (1981). Although actually being out-of-date, and in spite of the many inaccuracies contained in it, the most important comprehensive taxonomic work on the group still is Carlgren's survey on the families and genera (Carlgren 1949).
It is as yet impossible to make a reliable estimate of the number of species occurring on Indonesian reefs and other shallow-water habitats, but this number will probably not exceed a hundred.
The most conspicuous Actinaria on Indonesian reefs are species of Stichodactylidae, Thalassianthidae (fig. 8.19) and Actinodendronidae (fig. 8. 20). The members of these families are invariably symbiotic with zooxanthellae, and are often relatively large, and less contractile than other sea anemones. The largest species, viz. Stichodactyla mertensii and Heteractis magnifica, may reach a diameter of up to about 1 m across the oral disc when fully expanded. A host of organisms, such as shrimps, crabs, parasitic copepods (Humes 1982, 1985) and parasitic gastropods of the genus Epitonium (fig. 8.21) (e.g., den Hartog 1987), may be found associated with members of these families.
Perhaps most striking is the obligate association of the damselfish genera Amphiprion (27 species) and Premnas (1 species) (generally referred to as clownfishes or anemonefishes) with host anemones (about 10 species, generally referred to as clownfish anemones), mostly belonging to the Family Stichodactylidae. All species of host anemone recognized at present occur in Indonesia, and at least 14 of the known 28 species of anemone fish.
Associations between clownfishes and their host anemones vary from highly unspecific, as in the case of Amphiprion clarkii (which is known to associate with all species of host anemone, although some of these are clearly preferred to others), to mono-specific, as exemplified by Premnas biaculeatus (which associates exclusively with Entacmaea quadricolor) (colour plate 8.12).
The anemones were monographed by Dunn (1981) after the clownfish symbionts had been done by Allen (1972, 1974, 1980). A very nice recent field guide by Fautin and Allen (1992) combines and extends the data of both authors concerning distribution and host/symbiont combinations. During our expeditions in Indonesia, some interesting supplementary information of this sort was obtained. One detail, concerning the distribution of Amphiprion percula, may here be mentioned. This species, so far not recorded westward of Irian Jaya, was found to coexist with its twin species A ocellaris in Lembeh Strait, NE Sulawesi (id. confirmed by Dr. G.R. Allen). The distributional ranges of these species were so far thought to fully exclude each other.

Figure 8.17. A) Squash preparation showing tubes of ptychocysts, the building elements of cerianthid tubes. B) Detail of an empty ptychocyst capsule.
Photos by J.C. den Hartog.

Figure 8.18. Unidentified cerianthid with swarm of fish fry hovering above it.
Photo courtesy of W. Kolvoort, Pulau Pombo, Ambon, 1990.

Figure 8.19. Thalassianthus cf. senckenbergianus (Thalassianthidae); note the marginal globular nematospheres.
Photo by J.C. den Hartog, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.20. A) Actinodendron cf. plumosum, Spermonde Archipelago, SW Sulawesi. B) Small, unidentified species of Actinodendronidae, often found in Acropora-thickets.
A. Photo courtesy of B.W. Hoeksema, 1994; B. Photo by J.C. den Hartog, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.21. Epitonium sp. with egg-capsules; found associated with Heteractis crispa (Stichodactylidae).
Photo by J.C. den Hartog, 1994, Lembeh Strait, NE Sulawesi.
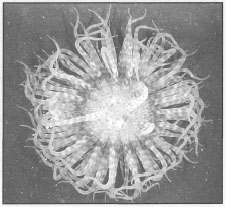
Figure 8.22. Boloceroides mcmurrichi (Boloceroididae), oral view of large specimen.
Photo by J.C. den Hartog, 1994, Spermonde Archipelago, SW Sulawesi.
The Family Actinodendronidae includes the genera Actinodendron, Megalactis and Actinostephanus. Actinodendrids are severe stingers which usually live in sheltered habitats with sandy or muddy bottoms, at depths ranging from 1 to 10 m (but also deeper). The column of the actinodendrids is buried deeply into the substratum, so that only the crown of branched tentacles is usually visible. Generally, these animals react to the slightest touch of the tentacles by sticking to and entangling the touching object or prey, and by simultaneously withdrawing body and tentacles into the substratum with a fast, swirling movement. When collecting actinodendrid anemones for scientific purposes, I generally use a spatulate iron bar (or lingis Ind.). Occasionally, failing to dig deep enough, the lower part of the body of an anemone may be pierced and severely damaged, surprisingly, however, without causing the animals to contract and withdraw. This seems to provide most interesting information about the nervous system of these anemones. An examination of food remains in the coelenteron of several specimens collected during the Indonesian-Dutch Snellius II Expedition (1984) and the Rumphius Biohistorical Expedition (1990) showed these to have consumed small fish. In this connection, it is of interest to note that expanded Actinodendron and Megalactis polyps often show a deceptive likeness to certain completely harmless soft corals, suggesting aggressive mimicry.
The genus Actinostephanus is represented by a single species, A. haeckeli. Owing to its long, unbranched arm-like tentacles with short protuberances and its colour, this peculiar anemone superficially resembles an octopod cephalopod. During the Rumphius Biohistorical Expedition, we therefore named the animal "Octopus anemone". This species was originally described from Ambon (Kwietniewski 1897), which is also the only locality where it has been collected so far (Roule 1909; Strack 1993). However, the species definitely has a wider distribution, for I have seen coloured slides of it from Flores (by Ms. L. Pitkin) and the Philippines (by Dr. G.C. Williams). In accordance with species of Actinodendronidae, the coelenteron of the "Octopus anemone" contained fish remains as well.
It may seem paradoxical that fish remains were also found incidentally in the coelenteron of the clownfish anemones Stichodactyla haddoni and Macrodactyla doreensis in Lembeh Strait in 1994. This suggests that the presence of symbiotic fish might in certain cases also have a decoy function, by luring other fish to entering these anemones. Surely, a study of both food and feeding behaviour of many of the large reef anemones may yield rewarding results.
An interesting habitat for Actiniaria is formed by Acropora and Montipora thickets, often found in very shallow water. These may harbour countless numbers of individuals, and a variety of species. Apart from characteristic species such as Boloceroides mcmurrichi (fig. 8.22), "Anemonia" manjano, Phymanthus sp. (fig. 8.23) and Thallassianthus cf. senckenbergianus, this habitat seems to provide a nursery for juvenile Stichodactylidae such as Heteractis crispa, H. aurora and Stichodactyla mertensii. These coral thickets may also shelter massive numbers of a small, bright-green, and as yet unidentified species of Actinodendronidae. Unlike its congeners, this species does not live in soft bottoms, but is loosely attached to the axils of coral branches. Since several specimens examined had well-developed gonads, it is unlikely that this population represents a juvenile brood of one of the larger species.

Figure 8.23. Phymanthus sp. (Phymanthidae), a species often encountered among Acropora thickets.
Photo courtesy of M.S.S. Lavaleye, 1990, Ambon; (c. x 0.6).

Figure 8.24. Mangrove mud flat at low tide in the inner Ambon Bay. Intertidal mud flats are an important habitat of Edwardsianthus pudica.
Photo by J.C. den Hartog, 1990.

Figure 8.25. Edwardsianthus pudica (Edwardsiidae) photographed at low tide in the inner Ambon Bay.
Photo by J.C. den Hartog, 1990, Ambon Bay.
Sandy and muddy substrates, as well as seagrass beds, are favourite habitats of sea anemones belonging to the infra-Order Athenaria, a group of predominantly non-zooxanthellate burrowing forms, comprising a number of families of doubtful relationship. Only a few, relatively conspicuous, species of this group were collected thus far in Indonesia. However, due to their often rather small size, burrowing way of life, and often secretive, nocturnal habits, athenarian anemones are often overlooked and may be far more common than assumed. A common zooxanthellate species found on mud flats in mangrove areas (e.g., inner Ambon Bay) (fig. 8.24) is Edwardsianthus pudica (Edwardsiidae) (fig. 8.25), which is fully expanded during daylight hours. Its 18-20 tentacles are always arranged in a very characteristic manner; the outer tentacles are spread on the bottom surface in groups of twos and threes, while the five inner tentacles are shorter and more or less erect with the tips often meeting above the centre of the oral disk.
Intertidal habitats, such as shallow tidal pools, moist cracks in substrate, holes and depressions in volcanic and/or coral rock, support a number of species including Anthopleura spp., Actinogeton spp. (both Actiniidae), Heteranthus verruculatus (Phymanthidae) and a small, somewhat deviating member of the Family Stichodactylidae, Stichodactyla tapetum.
Anemones of the infra-Order Boloceroidaria, notably of the Family Aliciidae, are well represented on Indonesian reefs. They are relatively mobile forms, which may easily move from place to place. Species of Boloceroides (Boloceroididae) are even able to swim by coordinated beating of their non-retractile tentacles; these are very easily shed, but have the ability to regenerate into new individuals. Two species are currently recognized, Boloceroides mcmurrichi (Central Indo-Pacific, including Indonesia) and B. hermaphoditica (Red Sea, east Africa), though on rather vague grounds (England 1992).
In members of the only non-zooxanthellate boloceroidarian genus Alicia, comprising several pantropical to subtropical species, the column is covered with numerous hollow cauliflower-shaped vesicles connected with the gastric cavity. The closely related zooxanthellate genus Bunodeopsis, in which are included rather similar, though much smaller, species, is known from the tropical/subtropical Pacific and Atlantic Oceans, but has not hitherto been recorded from Indonesia. Members of this genus might be found on the leaves of seagrass, among algae, on sponges, etc.
A very common attractive little species occurring throughout the Indo-west Pacific is Triactis producta (fig. 8.26). It reproduces prolifically by pedal laceration and is generally found in rather diffuse light conditions, under and between dead coral, among sponges, etc. The distal part of the column is characterized by branched outgrowths of the body wall, often referred to as pseudo-tentacles. These pseudo-tentacles bear whitish to bluish globular spherules representing extremely powerful nematocyst batteries. Very small specimens of this species, in which the pseudo-tentacles have not yet developed, are often carried by boxer crabs (Lybia spp.), one in each pincer (see fig. 8.26B).
Boxer crabs usually live hidden among coral rubble with algae, under stones, or among green calcareous algae such as Halimeda. Once detected these little crabs frantically move with their armed pincers towards potential aggressors.
Most interesting are members of the genus Phyllodiscus. These are the largest representatives of the Boloceroidaria. In accordance with members of the genus Alicia, their column is completely covered by compound vesicles. They may exceed 25 cm across the column with expanded vesicles. Species of Phyllodiscus are masters of camouflage and mimicry. Whether the genus actually represents a single, extremely variable species, Phyllodiscus semoni, or a number of specialists, each of which imitates another organism, is presently being studied (den Hartog and Hoeksema). It is most intriguing that this camouflage not only affects colour and pattern, but shape also. One form resembles variegated patches of algae-covered substratum. In another form, the columnar vesicles are long and ramified, the animal closely resembling a bunch of algae. Perhaps the most striking form is an almost perfect imitation of a colony of a branched coral (such as Acropora and Stylophora), the columnar vesicles closely resembling coral branches (fig. 8.27); the careful observer, however, will note that this imitation coral, unlike its rigid model, tends to move slowly to and fro, even in very calm water.

Figure 8.26. A) Triactis producta (Aliciidae), showing expanded oral disc and tentacles, and pseudo-tentacles provided with strongly developed globular nematocyst batteries. B) Boxer-crab Lybia tessellata with juvenile Triactis producta in its pincers.
A. Photo by J.C. den Hartog, 1992-1993, Seychelles; B. Photo courtesy of Ft. Steene.

Figure 8.27. Phyllodiscus sp. imitating substratum with short growth of algae; note symbiotic shrimps, Periclimenes holthuisi.
Photo courtesy of W. Kolvoort, 1994, Lembeh Strait, NE Sulawesi.

Figure 8.28. Aiptasia sp. (Aiptasiidae).
Photo courtesy of M.S.S. Lavaleye, 1990, Ambon.
The significance of this camouflage/mimicry in Phyllodiscus is not self-evident and needs clarification. Two possibilities are plausible as to the function of camouflage and mimicry: 1) to deceive potential prey, and 2) to distract potential predators. As species of Phyllodiscus do not normally expand their tentacles during the day, there seems little evidence in favour of the first possibility, so that the second possibility remains. However, considering that the vesicles of Phyllodiscus are provided with numerous globular, extremely powerful nematocyst batteries, one may wonder to which as-yet-unknown predators these anemones would be palatable (butterfly fishes?, hawksbill turtles?). Be that as it may, the combination of an almost perfect camouflage and the presence of powerful nematocyst batteries would seem to guarantee a better chance of survival than each of these two properties by itself. Having said this, these nematocyst batteries (also present in Triactis producta and the two species of the Caribbean genus Lebrunia) may not be functional primarily in the deterrence of potential predators, but rather in the competition for space with other sessile or semi-sessile reef organisms.
It is interesting to note that a clinical account by Hansen and Halstead (1971) on the stingings of a species referred to as "Hell's fire sea anemone" and identified as Actinodendron plumosum, actually bears on a species of Phyllodiscus, as is clear from the authors' descriptive notes and habitus and nematocyst photos. This does not alter the fact that the common name "Hell's fire sea anemone" could hardly have been chosen more appropriately (as I know from a marginal personal experience). The pain (and damage in the form of necrosis) suffered from contact with Phyllodiscus is primarily inflicted by the above-mentioned globular nematocyst batteries containing a type of nematocyst exclusively present in Boloceroidaria.
The large infra-Order Acontiaria is represented on Indonesian reefs by several small, less conspicuous or more secretive forms belonging to such genera as Aiptasia (fig. 8.28), Diadumene, Telmatactis (fig. 8.29) and related forms. The name 'Acontiaria' alludes to the most striking feature of this group, viz., the presence of acontia: defensive, heavily nematocyst-laden, thread-like organs springing from the free edge of the mesenteries, which are ejected through the mouth and/or permanent perforations of the body wall (cinclides) when an anemone is attacked or irritated.
Interesting forms of Acontiaria on account of their association with hermit crabs are Calliactis polypus and Verrillactis paguri. Individuals of these two species often occur together on the gastropod shell of a single hermit crab, C. polypus, located on top of the shell, V. paguri occupying the surroundings of the aperture (fig. 8.30). A similar situation exists in the Caribbean with regard to Calliactis tricolor and Verrillactis guttata (cf. Cutress and Ross 1969; as Sagartiomorphe guttata).
Another conspicuous acontiarian species on Indonesian reefs is the "Gorgonian wrapper" Nemanthus cf. annamensis (see Lavaleye and den Hartog 1995). This species, which is generally yellowish to orange variegated with dark specks or blots, can often be found in considerable numbers on gorgonians in water deeper than about 10 m (fig. 8.31). Although the genus Nemanthus is included in the Acontiaria, it actually deviates from all other members of this infra-Order by the absence of acontia proper. Instead, some of the mesenteries are provided with "acontioids" with very few nematocysts and an abundance of gland cells.

Figure 8.29. A) Telmatactis sp. (Isophelliidae), oral view. B) Telmatactis sp. side view.
Photos by J.C. den Hartog, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.30. Gastropod shell with Calliactis polypus (top and sides of shell) and Verrillactis paguri (spread around the aperture of the shell).
Photo by J.C. den Hartog, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.31. Nemanthus annamensis (Nemanthidae), the "Gorgonian wrapper".
Photo by J.C. den Hartog, 1994, Lembeh Strait, NE Sulawesi.
Order Zoantharia
Zoantharia form the second-largest group of sea anemones, presumably comprising a few hundred species, but contrary to the Actiniaria the diversity of the group is rather limited and most species are accommodated in only a few families and genera. The great majority of species are colonial, the polyps being connected by basal stolons or a common coenenchyme. A few species are solitary and free-living. Many species, notably of the genera Parazoanthus (Parazoanthidae) and Epizoanthus (Epizoanthidae), especially deeper-water forms, live associated with hermit crabs and sponges. Species of the dominant tropical shallow-water genera Zoanthus and Palythoa (both Zoanthidae) are always zooxanthellate (fig. 8.32).
Although members of these genera may be locally quite common in Indonesian shallow-water habitats, and sometimes very abundant, they are in my experience generally less dominant than in the tropical Atlantic. Our taxonomic knowledge of these genera is extremely poor and confused. Many of the numerous nominal species described from the Indo-Pacific are no doubt conspecific.
No one has probably published more on tropical Zoantharia than Pax. His papers mainly deal with Atlantic material, but he also published two major studies on species from Vietnam and one on species from Oceania (Pax and Muller 1956, 1957). A few of his papers contain descriptions of Indonesian material (Pax 1910, 1924). The most important classic studies on Indo-west Pacific species are those by Haddon and Shackleton (1891) and Haddon (1890) dealing with Torres Straits, and by Carlgren (1937), dealing with the Great Barrier Reef.
In recent years very few specialists have worked on the taxonomy of the group, and on a regional basis only. In the Indo-Pacific region, Walsh and Bowers (1971, 1977) studied the Hawaiian species, and Muirhead and Ryland (1993) presented a synopsis of the families and genera occurring on the Great Barrier Reef. In addition, Walsh (1967) published a useful attempt at a bibliography. A worldwide review of the genus Isaurus, though mainly based on material from Fiji, was published by Muirhead and Ryland (1985). It is interesting to note that these authors recognize three species only, one of which is I. tuberculatus, with a pantropical distribution. Several species of other genera undoubtedly are also widely distributed in the Indo-Pacific, and in conformity with Isaurus some may be distributed pantropically.
Truly encrusting forms, such as members of the genus Palythoa, are quite common on Indonesian coral reefs. Members of this genus are known particularly for their extremely poisonous properties due to the presence of palytoxine, a complicated compound that has been the subject of much research since it was discovered (see Mebst 1989; who also gives some references). One species, Palythoa toxica, was applied by the early Hawaiians to their arrow points to make them fatally poisonous (Walsh and Bowers 1971). Species of Palythoa are generally firm and tough, and well adapted to wave-exposed niches, and often occur intertidally. In the Atlantic and eastern Pacific, crabs of the genus Platypodiella are frequently found associated with members of this genus (den Hartog and Turkay 1991), but this phenomenon was never observed in Indonesian waters.
Members of the genus Zoanthus, soft-bodied and often very variable in form, thrive especially in calm, protected habitats, where they may be very common and form dense carpets, as, for example, at the eastern entrance of Lembeh Strait, NE Sulawesi. Well-known predators on Indo-Pacific zoanthids are gastropods of the genus Heliacus (Architectonicidae) (fig. 8.33) (Robertson 1967; Bieler 1993).
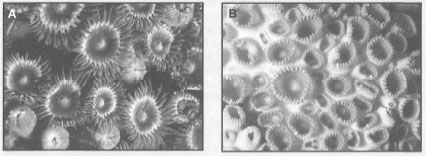
Figure 8.32. A) Zoanthus sp. Detail of colony with open and closed polyps. B) Palythoa cf. tuberculosa.
A. Photo courtesy of W. Kolvoort, 1994, Lembeh Strait, NE Sulawesi; B. Photo courtesy of R. Rozendaal, 1984, Sumba.

Figure 8.33. Zoanthus sp. with predatory gastropod Heliacus areola (Architectonicidae), a species occurring throughout the tropical Indo-Pacific. Note the peculiar cone-shaped operculum of the snail.
Photo courtesy of W. Kolvoort, 1994, Lembeh Strait, NE Sulawesi. Shells c. 15 mm in diameter.

Figure 8.34. Acrozoanthus australiae.
Photo courtesy of R. Rozendaal, 1984, Ambon Bay.
A common zoanthid inhabiting calm reefal habitats characterized by high sedimentation rates (e.g., Ambon Bay and Spermonde Archipelago, SW Sulawesi) is the atypical Acrozoanthus australiae, originally described from the Great Barrier Reef by Saville-Kent (1893). This species, redescribed by Ryland (in press), seems to prefer erect polychaete tubes as substratum, often forming dense colonies of rather large, long-tentacled green polyps, as a rule with a dark brown mouth and tentacle tips (fig. 8.34). Whether it also grows on other substrates, such as dead gorgonians, hydroids or antipatharians, needs verification.
To conclude this section, members of the most interesting Indo-Pacific genus Sphenopus (fig. 8.35) should not be omitted. They comprise a group of relatively large (up to at least 6 cm long and 4 cm wide), more or less tumbler-shaped, free-living solitary forms which live hidden in sandy and marl bottoms. Little is known about them.
Order Corallimorpharia
The Corallimorpharia form a small group of sea anemones which are very closely related to scleractinian corals. Indeed, whether they constitute a sister group of the Scleractinia or a polyphyletic group of skeletonless corals derived from several lineages of Scleractinia still is a matter of debate, but the most recent study on the subject (Fautin and Lowenstein 1993) seems to support the second opinion. The group comprises a few dozen species only. There are two main groups: the non-zooxanthellate Corallimorphidae and the zooxanthellate Discosomatidae and Ricordeidae. The first group is represented on Indonesian reefs by at least two species of Pseudocorynactis, viz. P. cf. caribbeorum and an as-yet-undescribed species (den Hartog in prep.) (fig. 8.36).
These species generally avoid bright daylight and live in caves, or hidden under blocks or plates of dead coral, under ledges, etc. The globular tentacle tips of these species contain the longest nematocysts and spirocysts known in Anthozoa. As spirocysts are functional in holding prey, this suggests that members of the genus Pseudocorynactis are formidable predators.
Members of the Family Discosomatidae are well-known, and more conspicuous reef elements. They are without exception clonal, exhibiting prolific asexual reproduction. The number of species is unknown, but my estimate is that some 10 to 12 species may occur on Indonesian reefs, some of which are still undescribed. In the field, it generally seems quite easy to recognize species, but once preserved the differences often seem much less clear. For this same reason opinions differ as to the number of genera to be recognized (den Hartog 1980). A taxonomic revision of the group is much needed. External morphology, never to be neglected in the taxonomy of sea anemones, is especially important in Discosomatidae, for the cnidoms of the various species prove quite uniform, whereas anatomical studies are thwarted by the flat disc-shaped form of the polyps, which makes it very difficult to make transverse sections. In contrast to members of the Corallimorphidae, the tentacles of all species of Discosomatidae lack spirocysts, a condition which is almost unique in sea anemones. Some species lack tentacles altogether. Therefore, speculating about the mode of feeding, one would be inclined to think that species of Discosomatidae are autotrophic and completely dependent on their symbiotic algae. However, Hamner and Dunn (1986) have shown how far such speculations may be beyond the truth. They provided evidence that species of Discosomatidae may feed at least facultatively by envelopment of prey, and even described a most interesting new species (Dunn and Hamner 1986) which proves to feed in this way upon fish! This species, Amplexidiscus fenestrafer (fig. 8.37), accommodated in a genus of its own, actually is a rather common species occurring throughout the Indo-west Pacific. Whether this feeding by envelopment is characteristic of all species of Discosomatidae remains to be seen; it would seem mechanically impossible in several of the more rigid forms such as species of Metarhodactis (fig. 8.38).

Figure 8.35. Sphenopus sp., a large, solitary and free-living zoanthid.
Photo by J.C. den Hartog, 1992, Seychelles.

Figure 8.36. Pseudocorynactis spec. nov. As yet undescribed, but not rare on Indonesian reefs.
Photo courtesy of B.W. Hoeksema, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.37. A) Amplexidiscus fenestrafer, expanded individual. B) Amplexidiscus fenestrafer, semi-closed individual (feeding by envelopment?).
A. Photo courtesy of R. Rozendaal, 1984, Tukang Besi Islands; B. Photo courtesy of B.W. Hoeksema, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.38. Large Metarhodactis sp. colony (i.e., clone) in very shallow water.
Photo courtesy of W. Kolvoort, 1990, Ambon Bay.

Figure 8.39. Spider crab, Cyclocoeloma tubercuolata, with associated discosmatid anemones; note eyes of crab.
Photo by J.C. den Hartog, 1994, Lembeh Strait, NE Sulawesi.

Figure 8.40. Discosoma (Rhodactis) cf. rhodostoma. Note marginal tentacles.
Photo courtesy of B.W. Hoeksema, 1994, Spermonde Archipelago, SW Sulawesi.

Figure 8.41. Photographs (A and B) of squash preparations of the marginal tentacles of Caribbean Discosoma sanctithomae, showing specialized nematocysts.
Photo by J.C. den Hartog.

Figure 8.42. Ricordea yuma, individual with single row of white tentacles.
Photo courtesy of R. Rozendaal, 1984, Ambon Bay.
Corallimorpharia, unlike Actiniaria, do not often form close associations with other organisms. A few parasitic copepods have been described (Humes 1982). However, as these small parasites are generally overlooked in the field, and because Corallimorpharia have never been systematically checked on their presence, there are undoubtedly more species to be discovered. There is one shrimp species that seems to be mainly restricted to species of Discosomatidae, Pliopontonia furtiva (Bruce 1973). A more manifest association is that between the spider crab Cyclocoeloma tuberculata and species of Discosomatidae (fig. 8.39). Whenever possible, this crab covers its carapace with discosomatid polyps (and in addition small colonies of Xeniidae are generally attached to the legs) (den Hartog 1990), as was also confirmed during recent expeditions to Ambon (1990) and North Sulawesi (1994). Being well camouflaged, the crab is generally found only when it moves, giving itself away by taking along its sessile discosomatid polyps.
Species of Discosomatidae, in accordance with many hard corals, may compete for space by extruding mesenterial filaments, thus killing and digesting neighbouring organisms. In addition, the tips of their marginal tentacles may develop nematocyst batteries containing numerous specialized nematocysts (cf. den Hartog 1977a) (figs. 8.40 and 8.41). At least some species are extremely aggressive and occasionally, notably in very shallow water, such species may be seen to form monocultures by prolific asexual reproduction, meanwhile killing and covering considerable patches of coral reef.
The most important difference between the monogeneric Family Ricordeidae and the Discosomatidae is the presence of spirocysts in the former and their total absence in the latter. The family is probably represented in Indonesian waters by only one species, Ricordea yuma. A peculiar feature of this species is the frequent presence of opaquely white tentacles, often arranged in a single radial row (fig. 8.42).
References.
Allen, G. R. (1972). The Anemonefishes: their Classification and Biology. T.F.H. Publications, Inc., Neptune City, New Jersey, 288 pp.
Allen, G. R. (1974). The Anemonefishes: their Classification and Biology (2nd Edition). T.F.H. Publications, Inc., Neptune City, New Jersey, 352 pp.
Allen, G. R. (1980). Anemonefishes of the World: Species, Care and Breeding. T.F.H. Publications, Neptune City, Inc., New Jersey, 104 pp.
Bieler, R. (1993). Architectonicidae of the Indo-Pacific (Mollusca, Gastropoda). Abh. naturw. Ver. Hamburg (NF) 30: 1-376.
Carlgren, O. (1900). Ostafrikanische Aktinien. Jb. hamb. wiss. Anst.17, Beih. 2: 23-144. Carlgren, O. (1924). Papers from Dr. Th. Mortensen's Pacific Expedition 1914-1916, XVI. Ceriantharia. Vidensk. Meddr dansk naturh. Foren. 75: 169-195
Carlgren, O. (1937). Ceriantharia and Zoantharia. Scient. Rep. Great Barrier Reef Exp. 19281929, 5(5): 177-207.
Carlgren, O. (1949). A survey of the Ptychodactiaria, Corallimorpharia and Actiniaria. Kungl. svenska Vetensk. Akad. Handl., ser. 4, 1(1): 1121.
Carlgren, O. (1950). Actiniaria and Corallimorpharia. Scient. Rep. Great Barrier Reef Exp. 1928-1929,5(7): 427-457.
Carlgren, O. (1956). Actiniaria from depths exceeding 6000 meters. Galathea Rep. 2: 916.
Cutress, C. E. and Ross, D. M. (1969). The sea anemone Calliactis tricolor and its association with the hermit crab Dardanus venosus. J. Zool. Lond. 158:225-241.
Dayton, P. K., England, K. W. and Robson, E. A. (in press). An unusual sea anemone, Dactylanthus antarcticus (Clubb 1908) (Order Ptychodactiaria), on gorgonians in Chilean fjords. In: den Hartog, J. C. et al. (eds.), Proceeding of the 6th International Conference on Coelenterate Biology, Noordwfkerhout, the Netherlands, 1995. National Natuurhistorisch Museum, Leiden.
Dunn, D. F. (1981). The Clownfish sea anemones: Stichodactylidae (Coelenterata: Actiniaria) and other sea anemones symbiotic with pomacentric fishes. Trans. Amer. Phil. Soc. Philadelphia: 3-115.
Dunn, D. F. and Hamner, W. M. (1987). Amplexidiscus fenestrafer n. gen., n. sp. (Coelenterata: Anthozoa), a tropical Indo-Pacific corallimorpharian. Micronesica 16(1): 29-36.
Emig, C. C. (1979). British and other phoronids. Synopsis of the British Fauna no 13: i-vi, 1 58.
England, K. W. (1987). Certain Actiniaria (Cnidaria, Anthozoa) from the Red Sea and tropical Indo-Pacific Ocean. Bull. Br. Mus. Nat. Hist (Zool.) 53(4): 205-292.
England, K. W. (1992). Actiniaria (Cnidaria: Anthozoa) from Hong Kong with additional data on similar species from Aden, Bahrain and Singapore. In: Morton, B. (ed.), The marine Flora and Fauna of Hong Kong and Southern China III, pp. 49-95. Hong Kong University Press.
England, K. W. and Robson, E. A. (1984). A new sea anemone from South Africa (Anthozoa, Ptychodactyaria). Ann. S. Afr. Mus. 94(5): 305-329.
Fautin, D. G. and Allen, G. R. (1992). Field Guide to Anemonefishes and their Host Sea Anemones. Western Australian Museum, Perth, 157 pp.
Fautin D. G. and Lowenstein, J. M. (1993). Phylogenetic relationships among scleractinian, actinians and corallimorpharians (Coelenterata: Anthozoa). Proc. 7th Int. Coral Reef Symp. Guam, 2: 665-670.
Haddon, A. C. (1898). The Actiniaria of Torres Straits. Scient. Trans. R. Dublin Soc. (2) 6(16): 393-498.
Haddon, A. C. and Shackleton, A. M. (1891). Reports on the zoological collections made in Torres Straits by professor A.C. Haddon, 1888-1889. Actiniae: I. Zoantheae. Trans. R. Dublin Soc. (2) 4(13): 673-701.
Hamner, W. M. and Dunn, D. F. (1986). Tropical Corallimorpharia (Coelenterata: Anthozoa): feeding by envelopment. Micronesica 16(1): 37-41.
Hansen, P. A. and Halstead, B. W. (1971). The venomous sea anemone Actinodendron plumosum Haddon of South Vietnam. Micronesica 7(1-2): 123-136.
Hartog, J. C. den (1977). Descriptions of two new Ceriantharia from the Caribbean region, Pachycerianthus curacaoensis n. sp. and Arachnanthus nocturnus n. sp., with a discussion of the cnidom and of the classification of Ceriantharia.— Zool. Med. Leiden 51(14): 211-242.
Hartog, J. C. den (1977a). The marginal tentacles of Rhodactis sanctithomae (Corallimorpharia) and the sweeper tentacles of Montastrea cavernosa (Scleractinia); their cnidom and possible function. Proc. 3rd Int. Coral Reef Symp., Miami: 463-469.
Hartog, J. C. den (1980). Caribbean shallow-water Corallimorpharia. Zool. Verh. Leiden 176: 1-83.
Hartog, J. C. den (1987). Observations on the wentletrap Epitonium clathratulum (Kanmacher, 1797) (Prosobranchia, Epitoniidae) and the sea anemone Bunodosoma biscayensis (Fischer, 1874) (Actiniaria, Actiniidae). Basteria 51: 95-108.
Hartog, J. C. den (1990). Associated occurrence of Cyclocoeloma tuberculata Miers, 1880 (Decapoda: Majidae) and species of Discosomatidae (Anthozoa: Corallimorpharia. Zool. Med. Leiden. 64: 161-168.
Hartog, J. C. den and Tukay, M. (1991). Platypodiella georgei spec. nov. (Brachyura: Xanthidae), a new crab from the island of St Helena, South Atlantic Ocean, with notes on the genus Platypodiella Guinot, 1967.
Humes, A. G. (1982). A review of Copepoda associated with sea anemones and anemonelike forms (Cnidaria, Anthozoa). Trans Amer. Phil. Soc. Philadelphia 72(2): 1-120.
Humes, A. G. (1985). Cnidarians and copepods: a success story. Trans Am. Microsc. Soc. 104(4): 313-320.
Klunzinger, C. B. (1877). Die Korallthiere des Rothen Meeres, pt. 1: Die Alcyonarien und malacodermen: i-vii, 1-98, pis 1-8. Verlag der Gutmann'schen Buchhandlung, Berlin.
Kwietniewski, C. R. (1896). Actiniaria von Ternate, nach den Sammlungen von Herrn Prof. Dr. W. Kukenthal. Zool. Anz. 19, 512: 388 391. Kwietniewski, C. R. (1897). Actiniaria von Ternate. Abhandl. Senckenb. naturforsch. Ges. 23:321-345. Kwietniewski, C. R. (1897a). Actiniaria von Ambon und Thursday Island. In: Semon, R. (ed.), Zoologische Forschungsreisen in Australien und in dem malayischen Archipel ausgeführt in dem Jahren 1891-1893, 5. Denkschr. med. -naturw. Ges. Jena 8: 385 430. Gustav Fisher, Jena.
Lavaleye, M. S. S. and Hartog, J. C. den (1995). A case of associated occurrence of the crab Lauridromia intermedia (Laurie 1906) (Crustacea: Decapoda: Dromiidae) and the actinian Nemanthus annamensis Carlgren, 1943 (Anthozoa: Actiniaria: nemathidae). Zool. Med. Leiden 69(11); 121-130.
Mariscal, R. N., Conklin, E. J. and Bigger, C. H. (1977). The Ptychocyst, a major new category of cnida used in tube construction by a cerianthid anemone. Biol. Bull. 152: 392-405.
McMurrich, J. P. (1910). The Actiniaria of the Siboga Expedition. Part 1. Ceriantharia. Siboga-expeditie 15a: 1-48.
Mebst, D. (1989). Gifte im Riff: Toxicologic und Biochemie eines Lebensraumes: 1-120. Stuttgart: Wiss. Verl.-Ges.
More R. E. and Scheuer, P. J. (1971). Palytoxin: a new marine toxin from a coelenterate. Science 172: 495-498.
Morton, B. (1989). Partnerships in the sea: Hong Kong's marine symbioses. Hong Kong University Press, Hong Kong and E.J. Brill, Leiden/New York/København/Köln, 124 pp.
Muirhead, A. and Ryland, J. S. (1985). A review of the genus Isaurus Gray, 1828 (Zoanthidea), including new records from Fiji. J. Nat. Hist. 19:323-335.
Muirhead, A. and Ryland, J. S. (1993). Order Zoanthidae (Class Anthozoa, Zoantharia). In: Mather, P. and Bennett, I. (eds.), A Coral Reef Handbook. Australian Coral Reef Society, Surrey Beatty & Sons Pty Limited, pp. 52-58.
Pax, F. (1910). Aktinien der Aru-Inseln. In: Merton, H. (ed), Ergebnissen zoologischen Forschungsreise südöstlich. Molukken (Aru und Kei-Inseln) 1. Abh. Senckenberg naturf. ges. 33: 297-304.
Pax, F. (1924). Anthozoen des Leidener Museums. Zool. Med. Leiden 8(1): 1-17.
Pax, F. and Muller, I. (1956). Zoantharien der "Mission Ranson en Oceanie" (1952). Mem. mus. hist. nat. ser. A, Zool. 8, 6: 225-247.
Pax, F. and Müller, I. (1957). Zoantharien aus Viet-nam. Mem. mus. national hist. nat. new ser., ser A, Zoologie 16, 1: 1-40.
Riemann-Zürneck, K. (1969). Cerianthus-Larven aus dem Weserästuar: Beobachtungen zur Biologie und Entwicklung. Veröff. Inst. Meeresforsch. Bremerh. 11: 37-46.
Robertson, R. (1967). Heliacus (Gastropoda: Architectonicidae) symbiotic with Zoanthiniaria (Coelenterata). Science 156(3772): 246-248.
Roule, L. (1909). Actiniaires d'Amboine. Rev. Suisse Zool. Geneve 17:113-120.
Ryland, J. S. (in press). Budding in Acrozoanthus Saville-Kent, 1892 (Anthozoa: Zoanthidae). In: den Hartog, J. C. et al. (eds.), Proceeding of the 6th International Conference on Coelenterate Biology, Noordwfkerhout, The Netherlands, 1995. National Natuurhistorisch Museum, Leiden.
Saville-Kent, W. (1893). The Great Barrier Reef of Australia: its products and potentialities. W. H. Allen and Co, London, 387 pp.
Strack, H. L. (1993). Results of the Rumphius Biohistorical Expedition to Ambon (1990). Part 1. General account and list of stations. Zool. Verh. Leiden 289: 1-72.
Walsh, G. E. (1967). An annotated bibliography of the families Zoanthidae, Epizoanthidae, and Parazoanthidae (Coelenterata, Zoantharia). Hawaii Inst. mar. biol. techn. rep. 13: 1-77.
Walsh, G. E. and Bowers, R. L. (1971). A review of Hawaiian zoanthids with descriptions of three new species. Zool. J. Linn. Soc. 50: 161-180.
Walsh, G. E. and Bowers, R. L. (1977). Order Zoanthiniaria. In: Devaney, D. M. and Eldredge, L. G. (eds.), Reef and Shore Fauna of Hawaii. Section 1: Protozoa through Ctenophora. Spec. Pubis Bernice P. Bishop Mus. 64(1): 148-157.